Catheter Stabilization Devices Market Size, Share Report 2026-2034
REPORT DETAILS
REPORT DETAILS
ABOUT THIS REPORT
Catheter Stabilization Devices Market Summary
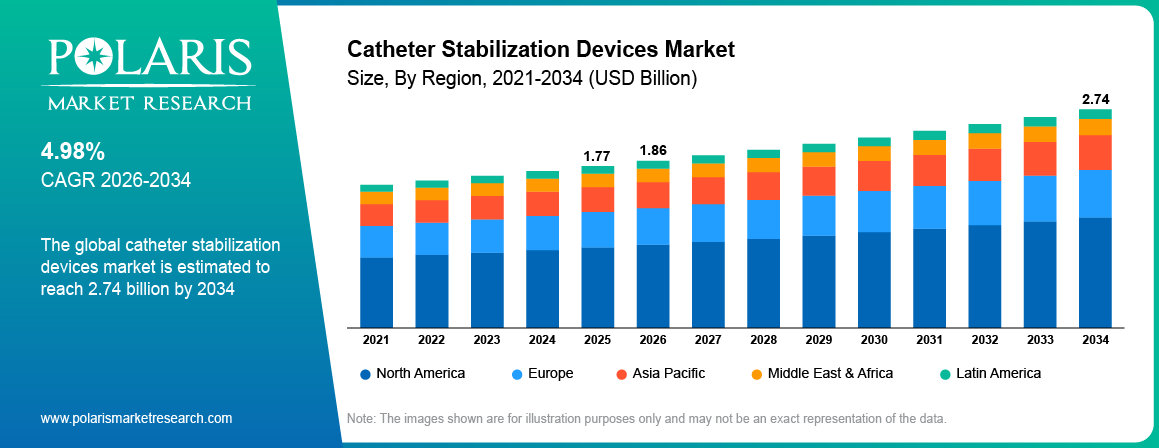
The global catheter stabilization devices market is estimated around USD 1.77 Billion in 2025,?with consistent growth anticipated during 2026–2034. This growth is driven by rising hospital procedures and stronger infection prevention focus that are increasing demand for catheter stabilization devices globally. The market is projected to grow at a CAGR of 4.98% during the forecast period.
Market Statistics
Key Takeaways
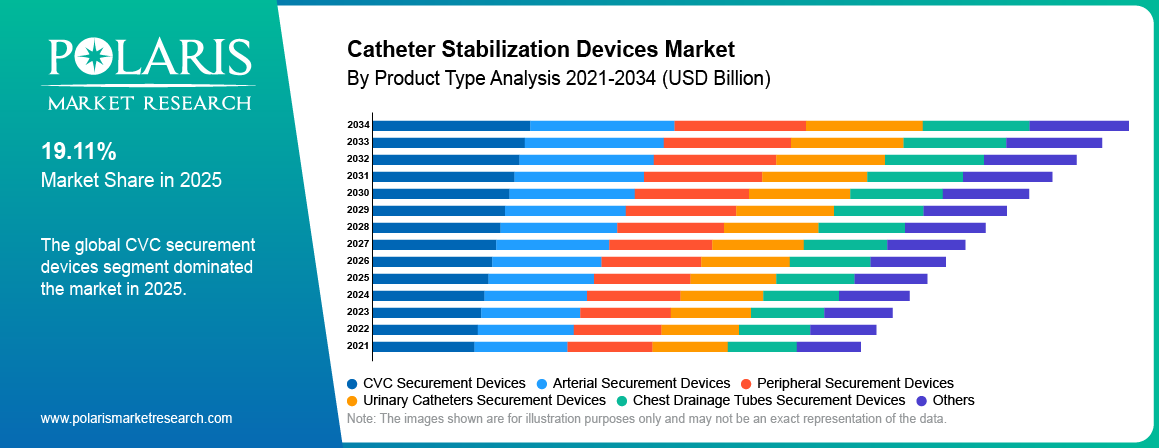
- The CVC securement devices segment was dominant with a revenue share of 29.00% in 2025 due to high use in critical care settings.
- The hospitals segment dominated with 68.00% share in 2025 due to large catheter procedure volumes.
- Rapid growth is expected at a CAGR of 6.59% in the home healthcare providers segment due to rising home infusion demand.
- Companies are focusing on expanding portfolios across vascular, urinary, and specialty securement products.
- Advanced adhesive and skin-friendly materials are improving product performance and market growth.
- North America dominated the market with a revenue share of 42.00% due to advanced healthcare infrastructure and strict safety standards.
Industry Dynamics
- Rising hospital admissions are increasing demand for catheter securement products.
- Stronger infection control focus is supporting wider product adoption globally.
- High product cost limits adoption in price-sensitive healthcare facilities.
- Product innovation is improving comfort, fixation strength, and clinical efficiency.
What is Catheter Stabilization Devices?
Catheter stabilization devices are medical products used to secure vascular and urinary catheters at the insertion site to reduce movement, accidental dislodgement, and migration. The products find application in hospitals, ambulatory clinics, and other health care facilities. The market growth is driven by the increasing need for safety and reduction in complications associated with catheterization within various health facilities.
Value chain for the industry comprises product development, procurement of raw materials, incorporation of adhesives technology, product validation, product approval, marketing and distribution, purchasing by hospitals, and after-sales services. The infection prevention recommendations by various bodies like the CDC and the Infusion Nurses Society are compelling the healthcare industry to incorporate advanced and secure device. Concern about minimizing catheter-associated bloodstream infection and urinary tract infections is driving the market.
For their use in medical applications, stabilization devices for central venous catheters, PICC lines, peripheral IV catheters, and urinary catheters are becoming increasingly popular. Use of these products in the healthcare setting is projected to increase patient satisfaction, reduce unnecessary catheter replacements, and facilitate service delivery. Innovations in adhesive technologies, antimicrobials, and release systems have had a positive impact on the design of catheter stabilization devices.
Drivers & Opportunities
Rising hospital admissions and surgical procedures are increasing demand for catheter stabilization devices: Growing visits by patients seeking surgeries, emergency treatment, and treatments for chronic diseases have been witnessed in healthcare institutions. According to Eurostat, the most frequently conducted surgery in the EU is that of cataracts with a total of 4.73 million surgeries conducted in 2022, while 1.10 million were cesarean deliveries.The aforementioned factors contribute towards rising numbers of catheterizations and catheter stabilization systems.
Growing focus on infection prevention is supporting adoption of advanced catheter securement products: Healthcare facilities are increasing efforts to prevent blood infections via catheters and UTIs. According to U.S. Centers for Disease Control and Prevention, in April 2024, correct handwashing practices, use of PPEs, sanitization, and safety precautions are key factors that need to be considered to prevent the occurrence of hospital acquired infections.Hence, medical practitioners have been integrating engineered stabilization devices to minimize catheter migration.
Restraints & Challenges
High product cost compared to adhesive tape is limiting adoption in price-sensitive hospitals: Engineered stabilizers have a greater unit price compared to conventional stabilizers that involve tapes. Budget-strapped public hospitals and small health-care facilities are still focused on lower-cost options. This factor is slowing wider penetration in cost-sensitive markets.
Opportunity
Growth in home healthcare is creating demand for easy-to-use stabilization devices: Home infusion therapy, elderly care, and long-term catheter management are expanding across many countries. There is now an increasing demand for easy-to-use and comfortable stabilizers among patients and caregivers. Thus, there is an emerging market for stabilizer makers.
Digital hospital procurement systems are improving product access for manufacturers: Hospitals are using e-procurement systems to facilitate their purchases and management of suppliers. In September 2025, Oracle launched AI-based solutions for the healthcare supply chain to ensure efficient management and procurement operations at lower costs for hospitals. These systems are helping manufacturers improve product visibility and strengthen institutional sales channels.

Segmental Insights
This report offers detailed coverage of the catheter stabilization devices market product type and end user to help readers identify the fastest expanding and most attractive demand segments.
By Product Type
-
CVC Securement Devices
Based on product type, the CVC securement devices segment dominated the market with 29.00% share in 2025, driven by high use of central venous catheters in critical care, oncology, and surgical settings. These devices are widely adopted in hospitals to reduce line movement and infection risk. Strong demand from acute care facilities supported segment growth.
-
Peripheral Securement Devices
Based on product type, the peripheral securement devices segment is projected to grow at the fastest CAGR of 4.70% during the forecast period, due to rising peripheral IV catheter placements in emergency rooms, outpatient centers, and day care settings. Increasing patient numbers and treatment cycles have enhanced product usage.
By End User
-
Hospitals
By end user type, the hospitals sector accounted for 68.00% share in 2025, owing to a large number of catheter procedures performed in inpatient wards, operating theaters, and intensive care units. In November 2025, Cathetrix introduced the product named Nephro-Safe in MEDICA 2025, which is a next-generation nephrostomy tube securement device that minimizes infection probability, accidental displacement, and discomfort of patients.
-
Home Healthcare Providers
Based on end user, the home healthcare providers segment is projected to grow at the fastest CAGR of 6.59%, during the forecast period, due to rising home infusion therapy, elderly care demand, and chronic disease management. Easy-to-use stabilization devices are supporting adoption in home settings.
Regional Analysis
North America Catheter Stabilization Devices Market Overview
North America held a large market share of 42.00%, due to strong hospital infrastructure, high adoption of infection prevention products, and strict healthcare quality standards. The US leads the region, supported by advanced acute care networks and broad use of catheter securement protocols. For instance, in March 2026, U.S. Food and Drug Administration (FDA) cleared Toro Neurovascular’s Toro 88 Superbore Catheter, a new large-bore stroke device designed to improve support, trackability, and stability in complex neurovascular procedures.
Asia Pacific Catheter Stabilization Devices Market Insights
Asia Pacific is expected to witness the fastest growth in terms of CAGR 6.83%, during the forecast period due to expanding hospital capacity, rising surgical volumes, and growing awareness of hospital-acquired infections in China, India, Japan, and South Korea. In December 2024, Stereotaxis announced China NMPA approval of the Magbot robotic magnetic navigation ablation catheter, expanding access to precision robotic treatment for complex cardiac arrhythmias. Moreover, China and Japan are major markets supported by large healthcare systems and stronger infection control standards. In India, private hospitals growth and rising requirements for high-quality healthcare services are contributing to the adoption of catheter stabilization systems.
Europe Market Insights
Europe has a considerable market share of 28.00%, owing to strong medical device regulations, established healthcare systems, and rising focus on patient safety. Countries such as Germany, France, and UK are major markets supported by high hospital procedure volumes and infection prevention programs. As per European Centre for Disease Prevention and Control, more than 3.5 million healthcare-associated infection cases occur annually across the EU/EEA, causing over 90,000 deaths and a major public health burden.This helps in supporting demand for advanced catheter securement products.

Competitive Landscape & Key Players
The catheter securement systems market is moderately fragmented, with stiff competition among international medtech corporations, vascular access device makers, and niche securement solutions providers. Product reliability, skin compatibility, infection prevention performance, regulatory compliance, and pricing remain key competitive factors in the market. Companies are focusing on portfolio expansion, advanced adhesive technologies, hospital partnerships, and growth in emerging healthcare markets.
The following are some of the leading companies in the industry 3M Company, Becton, Dickinson and Company, Baxter International Inc., Cardinal Health Inc., Convatec Group plc, ICU Medical Inc., Merit Medical Systems Inc., B. Braun SE, Medline Industries LP, Smiths Medical, Centurion Medical Products, Dale Medical Products Inc., and others.
Premium Insights
Infection Prevention is Strengthening the Value Case
Hospitals are placing higher focus on reducing catheter-related bloodstream infections and urinary tract infections through standardized securement practices. Unplanned catheter movement often increases replacement procedures, nursing workload, and treatment delays. Catheter stabilization devices help lower dislodgement risk, improve line integrity, and support better patient outcomes. Since healthcare providers are adopting the value-based care model, procurement teams have also taken a look at stabilization devices as an investment for enhancing the safety of patients rather than a mere expense.
Advanced Securement Technologies are Reshaping Product Demand
Key product innovations have been skin-friendly adhesives, antimicrobial securement pads, and catheters that do not require sutures. Such products help to minimize damage to the skin, increase patient comfort, and promote extended dwell times for catheters. High-end products not only minimize dressing changes but also make clinical operations more efficient. The advanced securement product category is smaller than the traditional product category, but its growth rate is faster.
Key Players
- 3M Company
- B. Braun SE
- Baxter International Inc.
- Becton, Dickinson and Company
- Cardinal Health Inc.
- Centurion Medical Products
- Convatec Group plc
- Dale Medical Products Inc.
- ICU Medical Inc.
- Medline Industries LP
- Merit Medical Systems Inc.
- Smiths Medical
Industry Developments
- In April 2026: Reflow Medical introduced the next generation of its Cora Flex and Cora Force torqueable microcatheters in the USA. These devices were specifically engineered for improved navigation, torqueability, and lesion crossing during difficult coronary procedures. [source: reflowmedical.com]
- In November 2025: Nephro-Safe, a new product by Cathetrix aimed at nephrostomy patients, was unveiled at MEDICA 2025. This tube stabilizer is developed to minimize the chances of infection, accidental removal, and patient distress. [source: prnewswire.com]
Catheter Stabilization Devices Market Segmentation
By Product Type Outlook (Revenue, USD Billion, 2021-2034)
- Arterial Securement Devices
- CVC Securement Devices
- Peripheral Securement Devices
- Urinary Catheters Securement Devices
- Chest Drainage Tubes Securement Devices
- Others
By End User Outlook (Revenue, USD Billion, 2021-2034)
- Hospitals
- Home Healthcare Providers
- Others
By Regional Outlook (Revenue, USD Billion, 2021-2034)
- North America
- US
- Canada
- Europe
- Germany
- France
- UK
- Italy
- Spain
- Netherlands
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Malaysia
- South Korea
- Indonesia
- Australia
- Vietnam
- Rest of Asia Pacific
- Middle East & Africa
- Saudi Arabia
- UAE
- Israel
- South Africa
- Rest of Middle East & Africa
- Latin America
- Mexico
- Brazil
- Argentina
- Rest of Latin America
Catheter Stabilization Devices Market Report Scope
|
Report Attributes |
Details |
|
Market Size in 2025 |
USD 1.77 Billion |
|
Market Size in 2026 |
USD 1.86 Billion |
|
Revenue Forecast by 2034 |
USD 2.74 Billion |
|
CAGR |
4.98% from 2026 to 2034 |
|
Base Year |
2025 |
|
Historical Data |
2021–2024 |
|
Forecast Period |
2026–2034 |
|
Quantitative Units |
Revenue in USD Billion and CAGR from 2026 to 2034 |
|
Report Coverage |
Revenue Forecast, Competitive Landscape, Growth Factors, and Industry Trends |
|
Segments Covered |
|
|
Regional Scope |
|
|
Competitive Landscape |
|
|
Report Format |
|
|
Customization |
Report customization as per your requirements with respect to countries, regions, and segmentation. |
FAQ's
The global market size was valued at USD 1.77 Billion in 2025 and is projected to grow to USD 2.74 Billion by 2034.
North America dominated the market with 42.00% share in 2025, due to strong hospitals and strict infection control standards.
Major applications include CVC lines, PICC lines, IV catheters, urinary catheters, and specialty lines.
A few of the key players in the market are 3M Company, Becton, Dickinson and Company, Baxter International Inc., Cardinal Health Inc., Convatec Group plc, ICU Medical Inc., Merit Medical Systems Inc., B. Braun SE, Medline Industries LP, Smiths Medical, Centurion Medical Products, Dale Medical Products Inc., and others.
Key drivers include rising admissions, more surgeries, and stronger infection prevention focus.
Major demand comes from hospitals, clinics, home healthcare, long-term care, and ambulatory centers.
The market outlook remains positive due to home care growth and product innovation.
Download Sample Report of Catheter Stabilization Devices Market
Please fill out the form to request a customized copy of the research report.
