Cervical Dysplasia Market Share, Size, Trends, Industry Analysis Report, 2025 - 2034
REPORT DETAILS
REPORT DETAILS
ABOUT THIS REPORT
Market Statistics
Market Overview
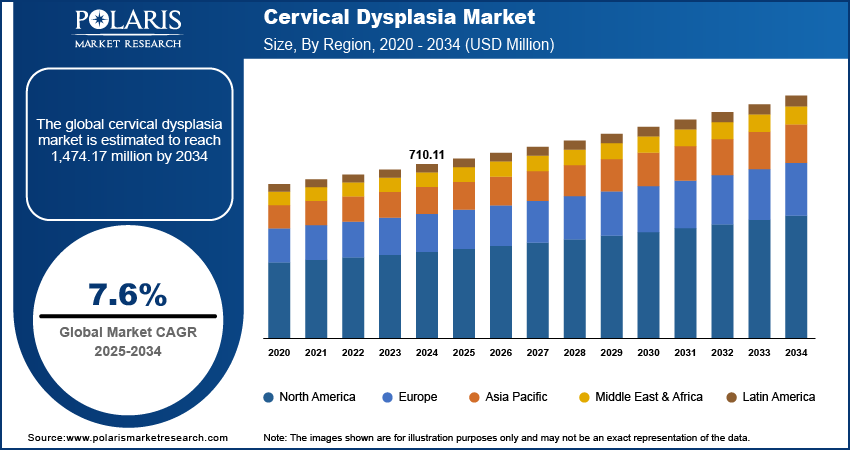
The global cervical dysplasia market was valued at USD 710.11 million in 2024 and is expected to grow at a CAGR of 7.6% during the forecast period. The growth is driven by rising prevalence of disease, rising government screening programs and technological advancement.
Key Insights
- The colposcope segment is projected to witness the highest growth rate due to the strategic collaboration between major players.
- In 2024, the hospital segment accounted for the largest share due to the high affordability and spending capacity of hospitals.
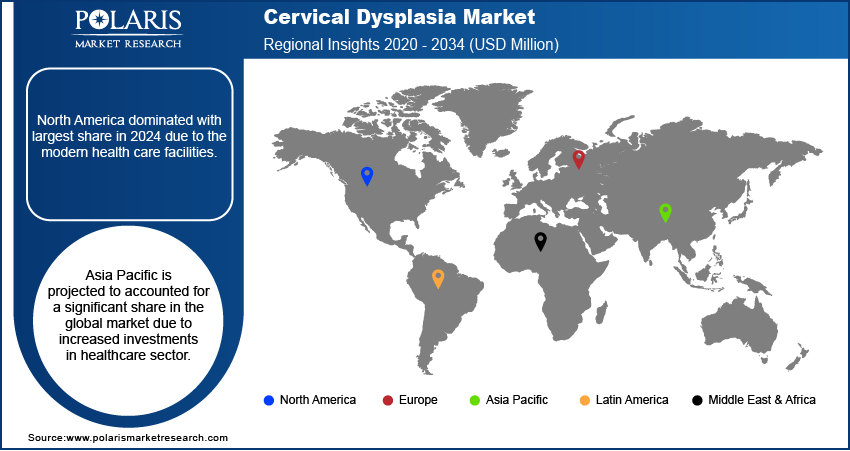
- North America dominated with largest share in 2024 due to the modern health care facilities.
- Asia Pacific is projected to accounted for a significant share in the global market due to increased investments in healthcare sector.
Industry Dynamics
- The government-funded screening programs is fueling the growth.
- The rising prevalence of disease is fueling the growth.
- Technological advancement is driving the growth.
- Limited awareness and screening access to low-income countries is limiting the growth.
Market Statistics
- 2024 Market Size: USD 710.11 Million
- 2034 Projected Market Size: USD 1,474.17 Million
- CAGR (2025-2034): 7.6%
- Largest Market: North America
To Understand More About this Research: Download Sample Report
Cervical dysplasia is an abnormal condition in which normal cells in the cervix region undergo abnormal changes. Cervix is the bottom part of the uterus, which finally leads into the vagina. This is the one, which dilates during childbirth, through which the fetus passes. In this condition, abnormal cells are not cancerous, but in later life, the stage could mutate into cancerous cells, if not treated or diagnosed on time. The Human Papillomavirus (HPV) is the main causal agent for cervical dysplasia.
The prevalence of cervical dysplasia is driving the demand for the effective therapeutics and diagnostics globally. According to the Columbian University, in U.S. alone, 250,000 to 1 million cases of cervical dysplasia are diagnosed every year. This rise in the cases are fueling the demand for the effective diagnosis. Rise in the HPV infection, smoking habits and others are driving these cases. Moreover, expanding healthcare infrastructure and healthcare access in urban as well as rural areas are boosting the number of identified cases which fuels the demand for the diagnostics and therapeutics of this disease, thereby driving the growth.
Industry Dynamics
Growth Drivers
The number of the government screening programs is rising worldwide. These screening programs are aimed to reduce or identify disease with low awareness among the general population, such as cervical dysplasia. This screening program is enabling the early detection of the disease, which in turn is fueling the demand for the diagnostic kits and medical devices which are required for this application. Moreover, rising screening programs in developing countries such as India, Mexico and Vietnam are further boosting the number of the identified cases and thereby fueling the growth of the industry.
Which Key Trends are Expected to Boost the Cervical Dysplasia Market During the Forecast Period?
The cervical dysplasia market is expected to experience significant transformation during the forecast period. Treatment modalities, advancements in diagnostics, and preventive strategies will drive it. Market players are focusing on adopting technological advancements and innovation. It helps them gain benefits in the competitive landscape. The following table consists of key future trends that are shaping the market:
| Trends | Benefits |
| Integration of Artificial Intelligence (AI) in Diagnostics
|
|
| Advancements in Molecular Diagnostics |
|
| Minimally Invasive Treatments |
|
| Personalized and Immunotherapy-Based Treatments |
|
| Expansion of At-Home Screening Solutions |
|

Cervical Dysplasia Market Report Scope
The market is primarily segmented on the basis of type, end-use, and region.
| By Type | By End-Use | By Region |
|
|
|
Know more about this report: Download Sample Report
Insight by Type
In 2024, the diagnostic tests market segment held the largest revenue share of the market for cervical dysplasia, among all. The factors responsible for this include positive reimbursement scenario, high efficacy, and cost-effectiveness. Its low cost resulted in higher penetration in emerging countries. Moreover, strong government initiatives also boosted the adoption of PAP smear tests. This had encouraged women to do smear tests to prevent future occurrences of the concerned condition.
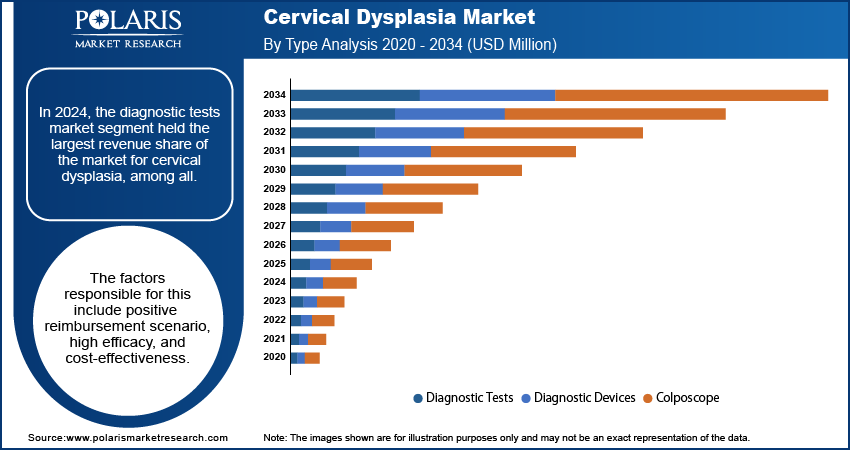
The colposcope market segment is projected to witness the highest growth rate of the market for cervical dysplasia over the assessment period. This future growth is attributed to the realizations in collaboration between market players. For instance, DYSIS Medical entered into an agreement with ITEM Medical Technologies to support the National Screening Programme for Turkish women in association with the Turkish Ministry of Health. Under this, the former company provided Colposcope cervical cancer screening. Thus, such growing initiatives are expected to benefit the market for cervical dysplasia in the near future.
Insight by End-Use
In 2024, the hospital market segment accounted for the largest share of the global cervical dysplasia market. This high share is attributed to the high affordability and spending capacity of hospitals. Moreover, the presence of favorable reimbursement scenarios and the emergence of specialty hospitals, which are better equipped with advanced devices also contributing to the cervical dysplasia industry segment’s growth prospects. Several hospitals in the developing region are undertaking various initiatives to carry out an early diagnosis, thereby fueling the segment growth.
Geographic Overview
Geographically, the global market for cervical dysplasia is bifurcated into North America, Asia Pacific, Europe, Latin America, and MEA. The North America region accounted largest market share in 2024. Regional factors boosting the market growth of cervical dysplasia include government-sponsored screening programs, a rise in the prevalence of associated diseases, and support initiatives for the adoption of HPV testing. The North America has one of the most modern healthcare infrastructure in urban as well as rural areas. This fuels the demand for the medical devices and diagnostic kits, thereby fueling the growth in North America region.
The Asia Pacific is expected to witness significant growth during the forecast period due to rise in the government investment in the healthcare sector. Governments across major developed and developing countries such as India, China and Japan are investing heavily in the healthcare sector. These investments are aimed to improve infrastructure and access to rural areas. Consequently, the number of healthcare facilities in region is rising which is boosting the detection of cervical dysplasia in urban and rural areas. This in turn is driving the demand for the medical devices and diagnostic kits required in cervical dysplasia, thereby driving the growth in the region.
Competitive Insight
The competitive landscape is characterized by continuous innovation in diagnostic technologies, increasing focus on minimally invasive procedures, and the integration of molecular testing methods. Companies are prioritizing research and development to enhance the accuracy and efficiency of HPV testing, colposcopy, and biopsy tools. Strategic collaborations with healthcare providers and government programs are common, aiming to expand screening reach and improve early detection. Market players are also investing in digital pathology and AI-driven analysis to support faster and more precise diagnosis. Intense competition exists in both developed and emerging markets, driven by rising awareness and screening initiatives.
Key Players
- Qiagen N.V.
- Abbott Laboratories
- Hologic, Inc.
- Quest Diagnostics
- DYSIS Medical Ltd
- Micromedic Technologies Ltd.
- OncoHealth Corporation, Inc.
- CooperSurgical Inc.
- Roche
Industry Developments
January 2025: Hologic completed the acquisition of Gynesonics for approximately USD 350 million, strengthening its women’s health portfolio with advanced minimally invasive fibroid treatment solutions.
May 2024: Roche gained FDA approval for its HPV self-collection solution, enabling early cervical cancer detection, supporting timely treatment, expanding its product portfolio, and creating new revenue opportunities.
May 2024: BD received FDA approval for a kit that allows self-collection of specimens for HPV testing. This approval seeks to broaden access to HPV screening, linked to cervical dysplasia and cervical cancer, adding value to BD’s business portfolio.
Cervical Dysplasia Market Report Scope
| Report Attributes | Details |
| Market Size in 2024 | USD 710.11 Million |
| Market Size in 2025 | USD 762.77 Million |
| Revenue Forecast by 2034 | USD 1,474.17 Million |
| CAGR | 7.6% from 2025 to 2034 |
| Base Year | 2024 |
| Historical Data | 2020–2023 |
| Forecast Period | 2025–2034 |
| Quantitative Units | Revenue in USD Million and CAGR from 2025 to 2034 |
| Report Coverage | Revenue Forecast, Competitive Landscape, Growth Factors, and Industry Trends |
| Segments Covered |
|
| Regional Scope |
|
| Competitive Landscape |
|
| Report Format |
|
| Customization | Report customization as per your requirements with respect to countries, regions, and segmentation. |
FAQ's
• The market size was valued at USD 710.11 Million in 2024 and is projected to grow to USD 1,474.17 Million by 2034.
• The market is projected to register a CAGR of 7.6% during the forecast period.
• A few of the key players in the market are Qiagen N.V., Abbott Laboratories, Hologic, Inc., Quest Diagnostics, DYSIS Medical Ltd, Micromedic Technologies Ltd., OncoHealth Corporation, Inc., CooperSurgical Inc., and Roche.
• The hospital accounted for the largest market share in 2024.
• The colposcope segment is expected to record significant growth.
Download Sample Report of cervical dysplasia market
Please fill out the form to request a customized copy of the research report.
