
Global eClinical Solutions Market [By Product & Solutions; By Deployment Type (On-Premises, Cloud Based); By Clinical Trial; By End Use; By Region]: Segments & Forecast, 2024 – 2032
- Published Date:Jan-2024
- Pages: 115
- Format: PDF
- Report ID: PM1093
- Base Year: 2023
- Historical Data: 2019-2022
Report Outlook
The Global eclinical solutions market was valued at USD 10.4 Billion in 2023 and is expected to grow at a CAGR of 14.10% during the forecast period. Global eClinical refers to using electronic technology in clinical trials and research. It involves the use of electronic data capture (EDC) systems, electronic patient-reported outcomes (ePRO) systems, and other electronic tools to streamline the clinical trial process. eClinical technology can help researchers and clinical trial managers to collect and manage data more efficiently, reduce errors, and speed up the development of new treatments.
Know more about this report: Request for sample pages
The use of eClinical technology has become increasingly popular in recent years due to its many advantages over traditional paper-based methods. eClinical technology allows data to be collected and stored in a centralized location, making it easier for researchers to access and analyze. This can reduce the time and cost associated with data management and analysis and improve data accuracy.
In addition to improving data management, eClinical technology can improve patient engagement and participation in clinical trials. By using ePRO systems, patients can easily report their symptoms and experiences, which can help to improve the quality of data collected. Electronic patient consent forms can also make the consent process more efficient and user-friendly for patients. eClinical technology can help to improve the overall efficiency of clinical trials. By automating certain processes, such as data entry and validation, eClinical technology can reduce the workload for researchers and clinical trial staff, allowing them to focus on other important tasks. Despite these advantages, there are also some challenges associated with the use of eClinical technology. One of the biggest challenges is ensuring the security and privacy of patient data. Researchers and clinical trial managers must ensure appropriate safeguards to protect patient information from unauthorized access or disclosure.

Know more about this report: Request for sample pages
Industry Dynamics
Growth Drivers
The clinical solution market is expected to grow significantly in the coming years, driven by several key factors. One of the primary growth drivers is the increasing demand for more efficient and cost-effective clinical trial processes. With the rising drug development costs and the complex regulatory landscape, pharmaceutical companies are looking for ways to accelerate clinical trial timelines and reduce costs without compromising quality.
Another key driver of eClinical growth is the expanding use of electronic health records (EHRs) and other digital health technologies. As more healthcare providers adopt EHRs and other digital technologies, it becomes easier to integrate patient data into clinical trials, improving data accuracy and reducing the burden on trial participants.
The COVID-19 pandemic has accelerated the adoption of eClinical solutions, as many clinical trials were disrupted or delayed due to social distancing measures and other restrictions. The need for remote patient monitoring and virtual clinical trials has driven the demand for eClinical solutions supporting these new clinical research models.
The growth driver for eClinical solutions is the increasing demand for personalized medicine. With technological advancements, it's now possible to analyze a patient's genetic profile and tailor treatments accordingly. This has led to a rise in clinical trials focused on personalized medicine, which requires more advanced data management and analysis tools.
eClinical solutions are the growing importance of data transparency and patient engagement. Patients increasingly demand more transparency in the clinical trial process, including access to their data. eClinical solutions can provide this by offering secure portals for patients to access their data and tools for researchers to analyze and share data more easily.
Report Segmentation
The market is primarily segmented based on products & solutions type, deployment, clinical trial, end use, and region.
|
By Products & Solutions Type |
By Deployment |
By Clinical Trial |
By End Use |
By Region |
|
|
|
|
|
Know more about this report: Request for sample pages
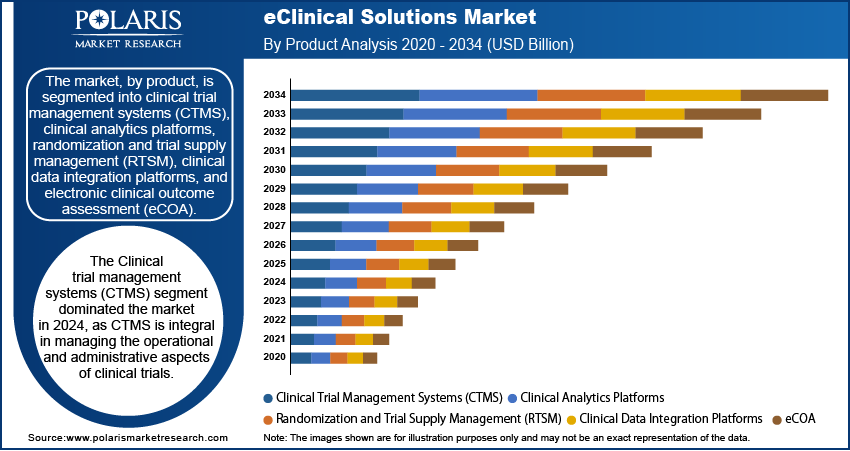
The Electronic Data Capture (EDC) and Clinical Data Management Systems (CDMS) segment is expected to witness the fastest growth
The market is expected to grow significantly in the coming years, particularly in the Electronic Data Capture (EDC) and Clinical Data Management Systems (CDMS) segment. EDC is a computerized system used to collect, manage, and report clinical trial data, while CDMS is a software application used to manage the data collected in clinical trials. The driver of growth in the eClinical solutions market is the increasing demand for clinical trial automation. Clinical trials are becoming more complex and time-consuming, leading to higher costs and longer timelines. EDC and CDMS solutions can streamline clinical trial processes, reduce errors, and improve data quality, reducing costs and shortening timelines. Another key growth driver in the eClinical solutions market is the increasing adoption of electronic health records (EHRs) and other digital healthcare technologies. As more healthcare organizations move towards digitalization, there is a growing need for tools to help manage and analyze the vast amounts of data generated by these systems. EDC and CDMS solutions can play an important role in this process, helping to ensure that data is accurate, reliable, and secure.
The Cloud-Based segment accounted for the largest market share in 2022
The global eClinical solutions market, which uses technology and software to manage clinical trials and related data in the healthcare industry, is expected to grow significantly in the coming years. One of the key drivers of this growth is the increasing adoption of cloud-based solutions.
Cloud-based eClinical solutions offer several advantages, such as cost-effectiveness, easy access to data, and scalability. With cloud-based solutions, clinical trial data can be accessed from anywhere, which is especially beneficial for remote clinical trials. Cloud-based solutions also offer greater flexibility in pricing models, allowing healthcare organizations to pay for only the services they require.
Another factor driving the adoption of cloud-based eClinical solutions is the need for greater collaboration and data sharing among stakeholders involved in clinical trials. Cloud-based solutions provide a centralized platform for stakeholders such as investigators, sponsors, and regulators to access and share data in real time, leading to more efficient clinical trial management.
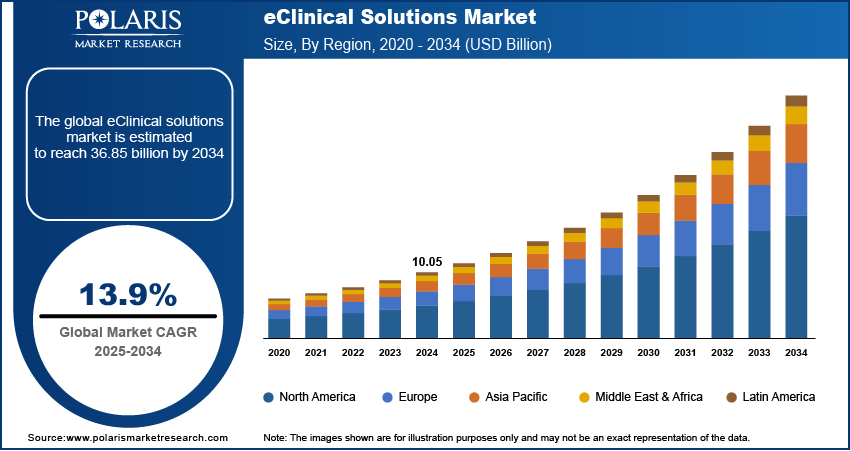
The demand in North America is expected to witness significant growth in 2022
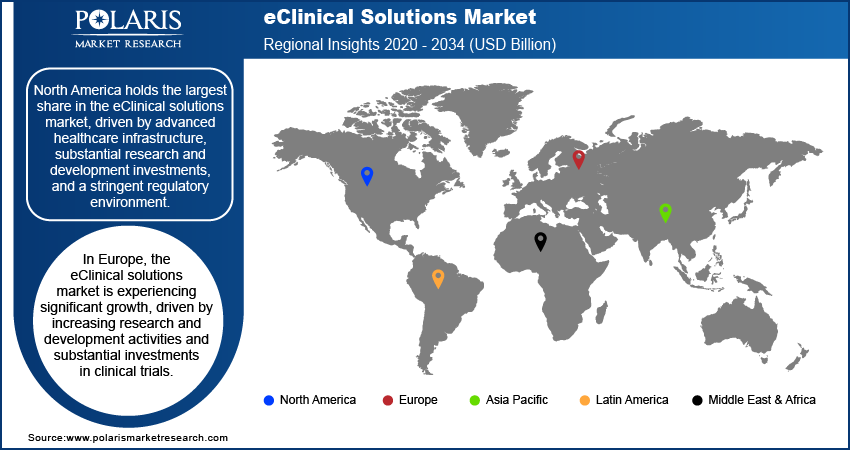
North America currently dominates the eClinical solutions market, followed by Europe. The Asia Pacific region is expected to witness the highest growth during the forecast period due to the increasing number of clinical trials conducted in the area.
North America currently dominates the eClinical solutions market, followed by Europe. However, the Asia Pacific region is expected to witness the highest growth during the forecast period due to the increasing number of clinical trials conducted in the area. The change in this region can be attributed to factors such as the large patient population, increasing R&D investments by pharmaceutical companies, and the growing adoption of technology in healthcare. Another factor driving the growth of the eClinical solutions market in North America is the increasing focus on precision medicine. Precision medicine involves the customization of medical treatment based on the individual characteristics of patients and requires the analysis of large amounts of patient data. EClinical solutions can help manage and analyze this data, making them a key component of precision medicine.
North America is also witnessing a shift towards value-based healthcare, emphasizing the value and outcomes of healthcare services rather than the volume of services provided. EClinical solutions can help healthcare providers measure and analyze results, making them an important tool in shifting toward value-based healthcare.
Competitive Insight
Some of the major players operating in the global market include CRF Health, DATATRAK International, Inc., Merge Healthcare Incorporated, ERT Clinical, eClinical Solutions LLC, Oracle Corporation, Medidata Solutions, Inc., Parexel International Corporation, BioClinica, Inc., OmniComm Systems, Inc.
Recent Developments
- April 2021 Datatrak International Introduces new launch of ePRO, eCOA, and eConsent to Help Advance Decentralized Trials Around the World.
- March 2023 Clario launches a cloud-based image viewer for clinical trials. Image Viewer is a comprehensive and easy-to-use solution for imaging needs in clinical trials. It offers high-resolution viewing and manipulation on demand in real time.
Global eClinical Solutions Market Report Scope
|
Report Attributes |
Details |
|
Market size value in 2024 |
USD 11.83 billion |
|
Revenue forecast in 2032 |
USD 33.95% billion |
|
CAGR |
14.10% from 2024 - 2032 |
|
Base year |
2023 |
|
Historical data |
2019 - 2022 |
|
Forecast period |
2024 - 2032 |
|
Quantitative units |
Revenue in USD billion and CAGR from 2024 to 2032 |
|
Segments covered |
By Products & Solutions Type, By Deployment, By Clinical Trial, By End User, By Region |
|
Regional scope |
North America, Europe, Asia Pacific, Latin America; Middle East & Africa |
|
Key companies |
CRF Health, DATATRAK International, Inc., Merge Healthcare Incorporated, ERT Clinical, eClinical Solutions LLC, Oracle Corporation, Medidata Solutions, Inc., Parexel International Corporation, BioClinica, Inc., OmniComm Systems, Inc. MedNet Solutions, PHT Corporation, DSG, Inc., Bio-Optronics, Inc., ArisGlobal LLC, Clincase GmbH, Anju Software, Inc., OpenClinica, LLC, Veeva Systems Inc., IBM Watson Health, Veeva Systems Inc. |
FAQ's
The eclinical solutions market report covering key segments are products & solutions type, deployment, clinical trial, end use, and region.
Global eClinical Solutions Market Size Worth $33.95 Billion By 2032.
The global eclinical solutions market expected to grow at a CAGR of 14.0% during the forecast period.
North America is leading the global market.
Key driving factors in eclinical solutions market are increased government grants to support clinical trials and growing adoption of software solutions in clinical research.

