NUT Midline Carcinoma Treatment Market Trends, Forecasts, and Key Players, 2026-2034
REPORT DETAILS
REPORT DETAILS
ABOUT THIS REPORT
NUT Midline Carcinoma Treatment Market Overview
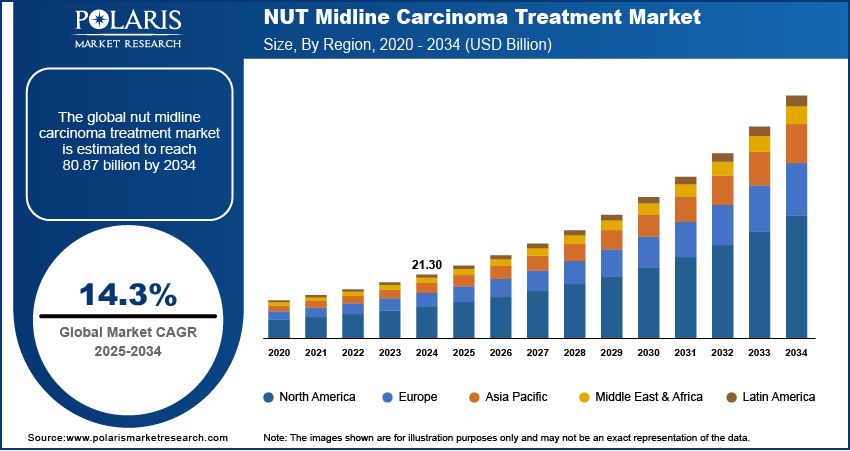
The global NUT midline carcinoma treatment market size was valued at USD 24.27 billion in 2025, growing at a CAGR of 14.3% from 2026 to 2034. Rising cancer incidence coupled with increasing healthcare expenditure is driving demand for NUT midline carcinoma treatment industry.
Key Insights
- North America led the global NUT midline carcinoma treatment market with a 39.43% revenue share in 2025. The regional market is primarily driven by expanding healthcare investments and growing molecular diagnostics adoption.
- Asia Pacific market is expected to grow at a 15.02% CAGR from 2026 to 2034. This is due to expanding healthcare investments and growing molecular diagnostics adoption.
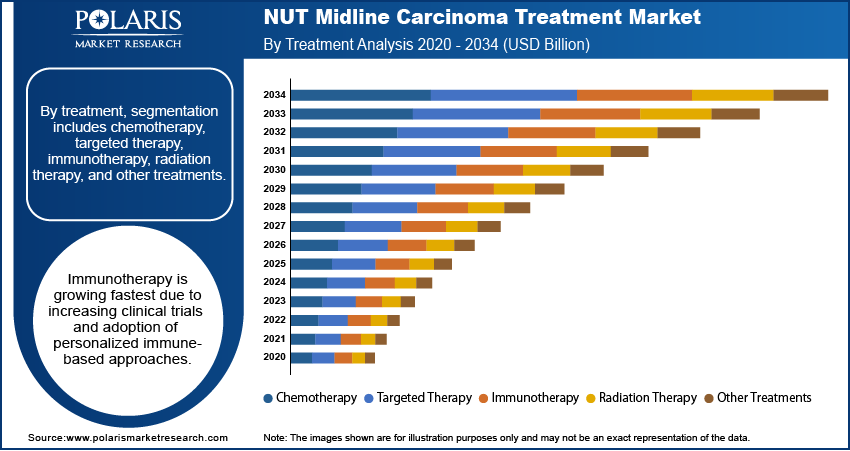
- The chemotherapy segment dominated with a 32.10% market share in 2025. This is owing to its capability to selectively inhibit NUTM1 gene fusions.
- The immunotherapy segment is estimated to grow at the highest CAGR of 16.6% throughout the forecast period based on rising clinical trials and the development of immune checkpoint inhibitors.
- The intravenous administration segment dominated with a 68.16% revenue share in 2025 due to its extensive application in delivering targeted and immunotherapy treatments effectively.
Note: Figures and projections outlined in this report are the result of Polaris Market Research’s proprietary analytical processes, grounded in the latest available datasets and market observations.
Industry Dynamics
- Rising cancer incidence worldwide is increasing demand for advanced oncology diagnostics in NUT midline carcinoma treatment.
- Growing healthcare expenditure across major economies supports adoption of precision cancer therapies.
- Development of BET inhibitors and other novel targeted agents addressing NUTM1 fusion pathways while creating opportunity in this market.
- Delayed diagnosis and limited disease awareness hinders early adoption of treatment.
Market Statistics
- 2025 Market Size: USD 24.27 Billion
- 2034 Projected Market Size: USD 80.87 Billion
- CAGR (2025–2034): 14.3%
- North America: Largest Market Share
The NUT midline carcinoma treatment market is aimed at therapies and diagnosis for rare and aggressive tumors caused by NUTM1 gene fusions. The treatments encompass molecular diagnostics, targeted therapies, and research-focused methods that target enhancing patient outcomes and early diagnosis. The market is comprised of pharmaceutical firms, diagnostic companies, and research institutes to develop efficient therapeutic strategies for this rare disease.
Rising global cancer incidence and increasing healthcare expenditure are driving demand for advanced oncology diagnostics. According to the American Medical Association, U.S. health spending rose 7.5% in 2023 to USD 4.9 trillion (USD 14,570 per person), higher than the 4.6% increase in 2022 and second only to the 10.4% rise in 2020. Increasing emphasis on targeted therapy and personalized medicine is further driving research and clinical use for NUT midline carcinoma. Growing partnerships among biopharma firms and academic centers are fueling the identification of new treatment pathways.
However, lack of disease awareness, low patient volumes, and prohibitive diagnostic expense limit broad adoption. The lack of approved targeted therapies and standardized diagnostic protocols remains an ongoing obstacle. Despite these barriers, ongoing genomic investigation and orphan drug incentives hold large opportunities for future market expansion.
Drivers & Opportunities
Global increase in cancer incidence: The rising incidence of cancer worldwide is creating a greater need for early and accurate diagnosis, which is driving demand for advanced oncology diagnostics in the NUT midline carcinoma treatment market. New cases of cancer are projected to increase to more than 35 million by 2050, an increase of 77% from an estimated 20 million in 2022, says the WHO. With more patients diagnosed with aggressive and rare forms of cancers, health providers are increasingly adopting to molecular and rare disease genetic testing technologies to facilitate early intervention as well as personalized treatment approaches.
Rising healthcare expenditure: Growing healthcare expenditures within developed and developing economies is driving the adoption of advanced cancer diagnostics and targeted medicines. Higher budgets allow hospitals, research centers, and biotech companies to invest in precision medicine infrastructure, molecular testing, and innovative treatment approaches, thereby expanding access to NUT midline carcinoma diagnostics and therapies.

Segmental Insights
NUT Midline Carcinoma Treatment Market Segmentation by Treatment
By treatment, the market is segmented into chemotherapy, targeted therapy, immunotherapy, radiation therapy, and other treatments. Among these, chemotherapy held the largest market share in 2025 owing to its ability to target the molecular mechanisms associated with NUTM1 gene fusions. With the shift towards precision cancer treatment, demand is likely to rise for both currently available and newly developed cancer treatments.
Chemotherapy
The chemotherapy segment dominated the NUT midline carcinoma treatment market with a 32.10% revenue share in 2025 and is anticipated to retain its leading position throughout the forecast period. Chemotherapy is one of the treatment options that holds significance, particularly as a first-line therapy and in combination with other therapies in cases where the cancer is either aggressive or has already metastasized. Chemotherapy is chosen when immediate action against the disease is required or when targeted therapy is not readily available. There has been a significant increase in the use of newer treatments, but chemotherapy continues to experience steady demand owing to its availability in many health care settings.
Targeted Therapy
Targeted therapies help inhibit the pathways involved in NUTM1 protein-fusion-driven cancers, making them a more targeted treatment option compared to conventional chemotherapies. BET inhibitors represent one of the most advanced targeted therapies that act upon cancerous tumors to stop their proliferation. The rising investments in clinical trials, orphan drugs, and biomarkers have fueled the development of the targeted therapies market segment.
Immunotherapy
The immunotherapy market segment is expected to witness the highest CAGR of 13.8% during the forecast period of 2026 to 2034, owing to increased clinical studies and the adoption of precision medicine in cancer treatment. The role of immune checkpoint inhibitors and next-generation immuno-oncology drugs is being explored for treating difficult-to-treat and rare cancers. Immunotherapy is becoming one of the most promising market segments amid rising requirements for long-lasting results and drug combinations.
Radiation Therapy
The application of radiation therapy remains relevant in localized tumor control, symptom management, and combination therapy. This modality is often used in conjunction with chemotherapy or surgery when necessary. Progress in radiation therapy technology is allowing for more precise administration and minimizing damage to surrounding tissues. Though not as large a segment in the market as systemic therapies, radiation therapy is a crucial component of NUT midline carcinoma management.
NUT Midline Carcinoma Treatment Market Segmentation by Route of Administration and End User
The NUT midline carcinoma treatment market is further segmented based on the route of administration and end users owing to the constant development of treatment techniques in oncology. The demand for easy modes of administration is increasing, and dedicated centers have enhanced access to advanced treatment modalities.
Route of Administration
Based on route of administration, the market is segmented into oral, intravenous (IV), and other. The intravenous (IV) segment led the market with a 68.16% revenue share in 2025, primarily due to the extensive use of this route for chemotherapy, immunotherapy, and several targeted cancer therapies available in clinical settings. IVs are preferred by hospitals and specialized facilities because they ensure that an exact dosage can be administered promptly and that adverse reactions can be monitored. Most established cancer therapy drugs continue to be administered using intravenous methods.
The oral segment is expected to experience rapid expansion at a 13.9% CAGR during the forecast period, largely driven by increasing patient demand for convenient therapy forms and fewer hospital visits. Growth in oral small-molecule drug forms, such as BET inhibitors and other targeted drugs, is also propelling segment growth.
End User
Based on end user, the market is segmented into hospitals, specialty clinics, and other end users. Hospitals held a leading market position with a 55.19% revenue share in 2025. This is due to multidisciplinary teams of oncologists, advanced imaging technologies, molecular diagnostic tests, and hospital-based treatment facilities. Hospital-based treatment facilities have always been considered the leading centers for the management of rare and aggressive cancers, which necessitates coordination between oncologists, pathologists, radiotherapists, and supportive care teams. The potential of hospitals to deliver complicated combination treatments will continue to contribute to their revenues.
The specialty clinics segment is expected to register the fastest growth at 14.1% CAGR during the forecast period. With the development of outpatient cancer treatment centers, specialty clinics are enhancing access to personalized care, follow-up care, and infusions conveniently. Increased awareness of rare cancers and decentralized cancer care are projected to drive this segment.
NUT Midline Carcinoma Treatment Market by Region
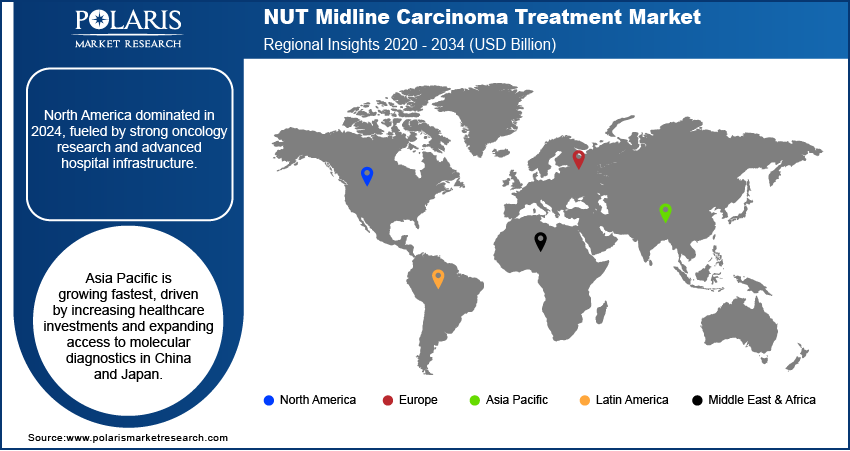
The global market for NUT midline carcinoma treatment varies according to the geographical regions, depending on the healthcare system, availability of molecular diagnostics, clinical trials, and knowledge of rare disorders. In 2025, North America accounted for the largest market share, whereas the Asia Pacific is expected to register the highest growth rate during the forecast period. Europe continues to contribute majorly to the revenues, while Latin America and MEA are improving in terms of treatment availability.
North America
The North America region accounted for the largest NUT midline carcinoma treatment market share of 39.43% in 2025. This is due to its well-equipped oncology facilities, substantial healthcare expenditure, and widespread uptake of precision medicine. This is attributed to the availability of genomics testing technology and the region’s involvement in rare cancers' clinical studies. The U.S. accounts for the largest regional market share. This is due to the presence of favorable policies for orphan drugs and pharmaceutical R&D. Additionally, there is increasing usage of biomarkers for therapeutic use in the country. Growing collaborations between cancer research centers and biotechnology firms are boosting market dynamics.
Asia Pacific
The Asia Pacific region is expected to register the highest CAGR of 15.02% from 2026 to 2034 due to increasing investments in healthcare infrastructure, rising awareness regarding rare types of cancers, and advancements in molecular diagnostics. The development of expertise in the areas of oncology, genomics, and biopharmaceuticals in countries such as China, Japan, and India is high. Government initiatives on cancer screening and personalized medicine are likely to boost market demand in the region.
Europe
The Europe region is anticipated to account for a considerable share of 15.48% during the forecast period owing to its developed healthcare infrastructure and growing popularity of customized treatments. In countries like Germany, the UK, France, and Italy, there is ongoing development of personalized medicine programs and approaches to rare disease treatment. Continuous research on cancer and international collaboration among clinics are driving market growth.
Latin America and Middle East & Africa
Latin America and the Middle East & Africa are also considered emerging regions where there is an improvement in accessibility to cancer diagnostics and facilities. Brazil, Mexico, Saudi Arabia, the UAE, and South Africa are among the major countries experiencing advances in their healthcare sectors. While these regions account for a smaller market share, there are future opportunities due to increasing health care expenditures and greater awareness of cancer.
Key Players & Competitive Analysis
The market for NUT midline carcinoma therapies is competitive with firms investing in the development of targeted therapies and increasing diagnostic capacity. Moreover, partnerships with multinational research institutions and pharmaceutical companies are driving clinical adoption and driving expansion in key markets.
Some of the large corporations in the treatment of NUT midline carcinoma include Merck & Co., Inc., Bristol-Myers Squibb Company, Pfizer Inc., F. Hoffmann-La Roche Ltd, C4 Therapeutics, Inc., Ipsen Biopharmaceuticals, Inc., GSK plc, Novartis AG, Syndax Pharmaceuticals Inc., ImageneBio Inc, Chimerix, NeoGenomics Laboratories, Zenith Epigenetics Ltd., and BioCentury Inc.
Key Players
- BioCentury Inc.
- Bristol-Myers Squibb Company
- C4 Therapeutics, Inc.
- Chimerix
- F. Hoffmann-La Roche Ltd
- GSK plc
- ImageneBio Inc
- Ipsen Biopharmaceuticals, Inc.
- Merck & Co., Inc.
- NeoGenomics Laboratories
- Novartis AG
- Pfizer Inc.
- Syndax Pharmaceuticals Inc.
- Zenith Epigenetics Ltd.
NUT Midline Carcinoma Treatment Pipeline and Recent Clinical Developments
ZEN-3694 Orphan Drug Designation
October 2025: Zenith Epigenetics revealed that the U.S. FDA granted Orphan Drug Designation to ZEN-3694, a novel oral BET inhibitor designed for NUT carcinoma. (source: zenithepigentics.com)
ZEN-3694 FDA Fast Track Designation
July 2025: Zenith Epigenetics’ ZEN-3694, an oral BET inhibitor, received FDA Fast Track Designation for treating metastatic or unresectable NUT midline carcinoma and is showing promising results in ongoing combination trials. (source: zenithepigentics.com)
Unmet Clinical Needs
The NUT carcinoma patient population has a life expectancy of about 6 months and there are currently no approved treatments for this condition, thus resulting in a significant unmet need which is fuelling an expedited approval process
Despite this condition affecting approximately 10,000 individuals on a yearly basis, it is underdiagnosed largely due to a lack of awareness and biomarker testing
NUT Midline Carcinoma Treatment Market Segmentation
By Treatment Outlook (Revenue, USD Billion, 2021–2034)
- Chemotherapy
- Targeted Therapy
- Immunotherapy
- Radiation Therapy
- Other Treatments
By Route Of Administration Outlook (Revenue, USD Billion, 2021–2034)
- Oral
- Intravenous (IV)
- Other
By End User Outlook (Revenue, USD Billion, 2021–2034)
- Hospitals
- Specialty Clinics
- Other End Users
By Regional Outlook (Revenue, USD Billion, 2021–2034)
- North America
- U.S.
- Canada
- Europe
- Germany
- France
- UK
- Italy
- Spain
- Netherlands
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Malaysia
- South Korea
- Indonesia
- Australia
- Vietnam
- Rest of Asia Pacific
- Middle East & Africa
- Saudi Arabia
- UAE
- Israel
- South Africa
- Rest of Middle East & Africa
- Latin America
- Mexico
- Brazil
- Argentina
- Rest of Latin America
NUT Midline Carcinoma Treatment Market Report Scope
|
Report Attributes |
Details |
|
Market Size in 2025 |
USD 24.27 Billion |
|
Market Size in 2026 |
USD 27.68 Billion |
|
Revenue Forecast by 2034 |
USD 80.87 Billion |
|
CAGR |
14.3% from 2026 to 2034 |
|
Base Year |
2025 |
|
Historical Data |
2021–2024 |
|
Forecast Period |
2026–2034 |
|
Quantitative Units |
Revenue in USD Billion and CAGR from 2026 to 2034 |
|
Report Coverage |
Revenue Forecast, Competitive Landscape, Growth Factors, and Industry Trends |
|
Segments Covered |
|
|
Regional Scope |
|
|
Competitive Landscape |
|
|
Report Format |
|
|
Customization |
Report customization as per your requirements with respect to countries, regions, and segmentation. |
FAQ's
The global market size was valued at USD 21.30 billion in 2024 and is projected to grow to USD 80.87 billion by 2034.
The global market is projected to register a CAGR of 14.3% during the forecast period.
North America led the market in 2024 due to advanced oncology infrastructure and widespread adoption of precision medicine technologies.
A few of the key players in the market are Merck & Co., Inc., Bristol-Myers Squibb Company, Pfizer Inc., F. Hoffmann-La Roche Ltd, C4 Therapeutics, Inc., Ipsen Biopharmaceuticals, Inc., GSK plc, Novartis AG, Syndax Pharmaceuticals Inc., ImageneBio Inc, Chimerix, NeoGenomics Laboratories, Zenith Epigenetics Ltd., and BioCentury Inc.
Targeted therapy dominated in 2024, driven by its ability to specifically inhibit NUTM1 gene fusions and improve patient outcomes.
Specialty clinics is growing steadily due to expansion of oncology-focused outpatient centers and rising adoption of personalized cancer therapies.
Asia Pacific is witnessing rapid growth due to increasing investments in healthcare infrastructure and rising awareness regarding rare types of cancers.
ZEN-3694 is an oral, small-molecule BET bromodomain inhibitor developed by Zenith Epigenetics. The FDA has granted both Orphan Drug Designation (October 2025) and Fast Track Designation (July 2025) to ZEN-3694 for the treatment of NUT carcinoma.
NUT carcinoma and NUT midline carcinoma (NMC) are generally the same, extremely aggressive cancer, with "NUT carcinoma" being the updated, preferred term.
BET inhibitors (BETi) are targeted treatments that interfere with the binding of BRD4 proteins to DNA, thus blocking the expression of the oncogenes that drive the carcinogenesis process.
Download Sample Report of NUT Midline Carcinoma Treatment Market
Please fill out the form to request a customized copy of the research report.
