Single-Use Filtration Assemblies Market Size, share, Growth Report, 2026-2034
REPORT DETAILS
REPORT DETAILS
ABOUT THIS REPORT
Single-Use Filtration Assemblies Market Summary
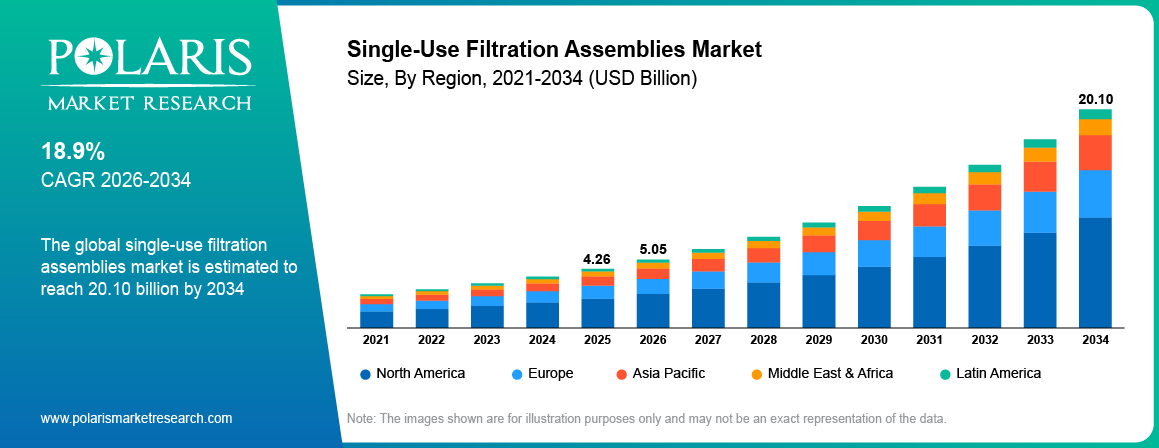
The global single-use filtration assemblies market is estimated around USD 4.26 Billion in 2025,with consistent growth anticipated during 2026–2034. This growth is driven by rising biologics production and expanding vaccine manufacturing that are increasing demand for single-use filtration assemblies globally. The market is projected to grow at a CAGR of 18.9% during the forecast period.
Market Statistics
Key Takeaways
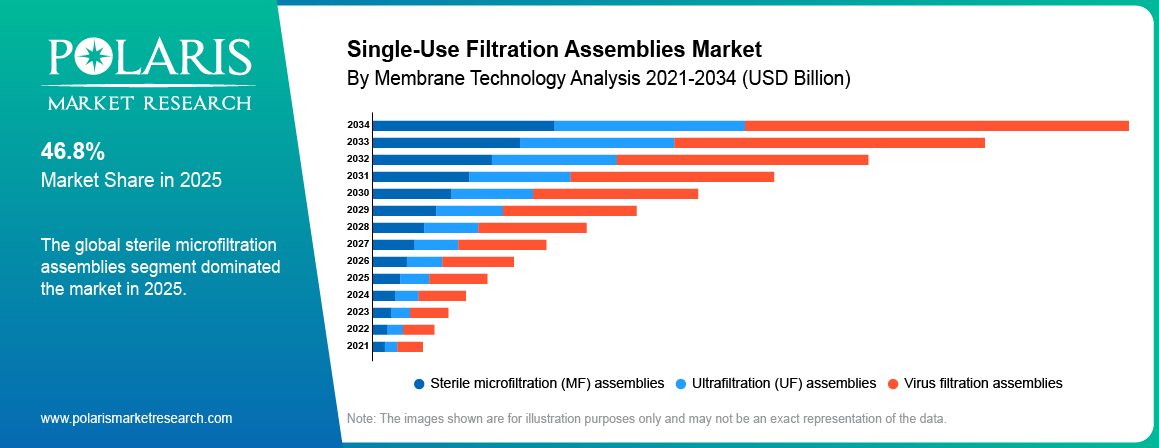
- Sterile microfiltration assemblies segment was dominant by 46.8% share in 2025 due to high use in sterile processing applications.
- Monoclonal antibody manufacturing segment dominated by 43.2% share in 2025 due to large commercial biologics production volumes.
- Rapid growth is expected in the gene and cell therapy segment with the fastest growth rate of 19.3% due to rising advanced therapy development.
- North America dominated the market with 39.38% revenue share due to advanced biomanufacturing infrastructure and strong regulatory standards.
- Asia Pacific is the fastest-growing region in the single-use filtration assemblies market, growing at a CAGR of 19.5% owing to expanding biologics manufacturing and increasing vaccine production.
Industry Dynamics
- Rising biologics production is increasing demand for disposable filtration systems.
- Growth of vaccine manufacturing is supporting higher use of ready-to-use assemblies.
- High recurring consumable costs are limiting adoption in price-sensitive facilities.
- Gene Therapy and Advanced Therapy Medicinal Products (ATMPs) are creating opportunity for specialized filtration assemblies.
What is Single-Use Filtration Assemblies?
Single-use filtration assemblies are pre-assembled disposable systems used in biopharmaceutical manufacturing to support sterile filtration, clarification, concentration, and fluid transfer processes. These products are made up of filtration membranes, tubes, fittings, manifolds, and housings, thus forming an integrated product. These are common in biologics plants, vaccine plants, and contract manufacturing organizations that have a need for speed and efficiency. Growth in market is driven by demand for flexible manufacturing solutions and reduction in contamination possibilities.
The industrial value chain consists of design, procurement of membranes, manufacturing of polymer parts, assembly, sterilization, testing, regulatory compliance, distribution, and application in production plants. Increasing interest in minimizing the cleaning-validation period and conserving water and energy is leading manufacturers to adopt a disposable approach. Adoption of products is also boosted by growing regulatory acceptance of validated single-use technology.
Single use filtration assemblies are extensively applied in the production of monoclonal antibodies, cell and gene therapies, vaccines, and buffers. Single use filtration assemblies play a vital role in optimizing batch turnover, mitigating contamination risks, and enhancing process flexibility. Continued development in high flow membrane materials, leak-free connections, and scalable filtration technologies is contributing to growth opportunities in the market.
Drivers & Opportunities
Rising Biologics Production is Increasing Demand for Disposable Filtration Systems: The biopharmaceutical companies are scaling up the manufacture of monoclonal antibodies, recombinant proteins, and biosimilar products across all the regions worldwide. According to EFPIA 2025 report, the value of pharmaceutical manufacturing reached USD 517 billion in 2024 from about USD 379 billion in 2020, which indicates the consistent increase in global demand for medicines along with manufacturing activities. Such type of molecules need sterile and effective filtration process during both upstream and downstream processing.
Growth of Vaccine Manufacturing is Supporting Higher Use of Ready-to-use Assemblies: The manufacturers of vaccines are enhancing their manufacturing capability tDo cater to both normal and emergency requirements related to vaccination. According to Global Health Press, the total output of vaccines in the world is approximately 14.5 billion doses per year, with the market for vaccines having grown close to USD 35 billion between 2024 and 2025. Therefore, prefabricated filtration units enable reduced setup time and rapid changeover of batches.
Restraints & Challenges
High Recurring Consumable Costs are Limiting Adoption in Price-Sensitive Facilities: The single use filter assemblies need to be replaced after every cycle, leading to an increase in operational costs in the long run. Smaller companies and facilities with budget constraints are hesitant about implementing them on a large scale. This trend presents a barrier to widespread adoption in the market.
Opportunity
Gene Therapy and Advanced Therapy Medicinal Products (ATMPs) are Creating Demand for Specialized Filtration Assemblies: Sterility of processing, flexibility in batch sizes, and safeguarding of products are essential in gene therapy and advanced therapy medicinal products (ATMP) manufacturing processes. In December 2025, Foxx Life Sciences introduced EZcellGENE single-use assemblies for gene therapy applications. As single-use filtration systems support these needs through closed and scalable operations.

Segmental Insights
This report offers detailed coverage of the single-use filtration assemblies market membrane technology and application to help readers identify the fastest expanding and most attractive demand segments.
By Membrane Technology
-
Sterile microfiltration (MF) assemblies
Based on membrane technology, the sterile microfiltration assemblies segment was dominant by 46.8% share in 2025, driven by high use in sterile filtration of media, buffers, and final fill processes. These assemblies are widely adopted across biologics and vaccine production sites. Strong demand for contamination control supported segment growth.
-
Virus filtration assemblies
Based on membrane technology, the virus filtration assemblies segment is projected to grow at the fastest CAGR during the forecast period, due to rising regulatory focus on viral safety in biologics manufacturing. Increased production of monoclonal antibodies and advanced therapies has expanded product demand.
By Application
-
Monoclonal antibody (mAb) manufacturing
Based on application, the monoclonal antibody manufacturing segment dominated by 43.2% share in 2025, driven by large-scale global production of antibody therapies for cancer and autoimmune diseases. These processes require multiple filtration stages across production cycles. High commercial output supported segment leadership.
-
Gene and cell therapy applications
Based on application, rapid growth is expected in the gene and cell therapy segment with the fastest growth rate of 19.3% during the forecast period, due to rising clinical pipeline activity and growing commercialization of advanced therapies. Small batch processing needs are increasing adoption of flexible single-use filtration systems.
Regional Analysis
North America Single-Use Filtration Assemblies Market Overview
North America dominated the market with 39.38% revenue share due to strong biologics manufacturing capacity, high adoption of disposable bioprocessing systems, and advanced regulatory standards. The US leads the region, supported by major monoclonal antibody producers and expanding CDMO operations. In April 2026, Freudenberg Medical introduced the ISO Class 5 cleanroom known as CleanAssure, which is used to assemble sterile single-use components, thus increasing validation and improving supply security for biopharmaceutical manufacturing processes.
Asia Pacific Single-Use Filtration Assemblies Market Insights
Asia Pacific is the fastest-growing region in the single-use filtration assemblies market, growing at a CAGR of 19.5% due to rapid expansion of biopharmaceutical production, rising biosimilar approvals, and growing CDMO capacity in China, India, Japan, and South Korea. China leads the region with strong domestic biologics output and new manufacturing sites. In India, government support for local biopharma production and rising private investments are increasing adoption of single-use filtration systems. As per the Indian Ministry of Chemicals and Fertilizers 2026 report, the Government launched the USD 1.06 billion Biopharma SHAKTI scheme to expand India’s biologics and biosimilars ecosystem and strengthen its global biopharma hub position.
Europe Market Insights
Europe has a considerable market share owing to strong pharmaceutical manufacturing infrastructure, strict sterility regulations, and rising investment in advanced therapies. Countries such as Germany, Switzerland, Ireland, and UK are major markets supported by established biologics plants and vaccine production facilities. For instance, in March 2026, Moderna announced the opening of its mRNA facility in the UK, which has the capacity to produce up to 100 million doses per year. In addition, the updated EU GMP Annex 1 standards are increasing focus on contamination control, which is supporting demand for single-use filtration assemblies.
Rest of the World Market Insights
Rest of the World holds a developing market share due to rising investments in domestic biopharmaceutical manufacturing and vaccine self-sufficiency programs. There are countries that have been increasing their capacity for producing biological products and vaccines. In April 2025, Boston Oncology is planning to invest USD 70 million to increase biomanufacturing capacity in Saudi Arabia. It will add sterile injectable filling lines, thus ensuring an adequate supply of biologics, biosimilars, and complex injectables in the region. The rising use of advanced bioprocessing facilities has boosted the need for single-use filtration assemblies in Latin America, the Middle East, and Africa.

Competitive Landscape & Key Players
The market for single-use filtration systems is moderately fragmented, with robust competition existing among bioprocess equipment manufacturers, filtration technology vendors, and single-use system suppliers. Product quality, sterility assurance, filtration efficiency, regulatory compliance, and pricing remain key competitive factors in the market. Companies are focusing on portfolio expansion, high-performance membrane technologies, strategic partnerships with biopharma manufacturers, and expansion in emerging bioprocessing markets.
The following are some of the leading companies in the industry Merck KGaA, Danaher Corporation, Sartorius AG, Thermo Fisher Scientific Inc., Parker-Hannifin Corporation, Repligen Corporation, Donaldson Company Inc., Meissner Filtration Products Inc., Asahi Kasei Corporation, 3M Company, Eaton Corporation plc, Avantor Inc., and others.
Premium Insights
- Integrated portfolio advantage: Biopharma manufacturers are reducing supplier complexity and preferring vendors that offer complete filtration solutions across sterile filtration, ultrafiltration, virus filtration, and fluid management systems. This trend is increasing competitive pressure on niche suppliers and supporting strategic acquisitions.
- Custom assembly demand growth: There is increasing demand for customized filtration assemblies tailored to specific processes that are integrated directly into bioreactor, mixing, and chromatography systems. This trend benefits from rising pricing leverage and long-term supply agreements.
- High-value advanced therapy segment: Gene therapy and cell therapy products will need high-end filtration technologies that offer greater sterility, less product loss, and batch processing. The margins on such products are relatively high even though the volume currently is lower compared to other markets. Early partnerships in advanced therapy production are creating long-term revenue opportunities.
Key Players
- 3M Company
- Asahi Kasei Corporation
- Avantor Inc.
- Danaher Corporation
- Donaldson Company Inc.
- Eaton Corporation plc
- Meissner Filtration Products Inc.
- Merck KGaA
- Parker-Hannifin Corporation
- Repligen Corporation
- Sartorius AG
- Thermo Fisher Scientific Inc.
Industry Developments
- In April 2026: Avantor has made an announcement regarding the extension of its Masterflex product line in order to improve fluid handling from research and development to manufacturing processes. [source: ir.avantorsciences.com]
- In April 2026: Saint-Gobain Life Sciences and VERDOT partnered to launch integrated PUPSIT solutions for bioprocessing, improving sterility compliance, automation, and GMP-ready filtration workflows. [source: saint-gobain.com]
Single-Use Filtration Assemblies Market Segmentation
By Membrane Technology Outlook (Revenue, USD Billion, 2021-2034)
- Sterile microfiltration (MF) assemblies
- Ultrafiltration (UF) assemblies
- Virus filtration assemblies
By Application Outlook (Revenue, USD Billion, 2021-2034)
- Monoclonal antibody (mAb) manufacturing
- Vaccine manufacturing
- Gene and cell therapy applications
- Others
By Regional Outlook (Revenue, USD Billion, 2021-2034)
- North America
- US
- Canada
- Europe
- Germany
- France
- UK
- Italy
- Spain
- Netherlands
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Malaysia
- South Korea
- Indonesia
- Australia
- Vietnam
- Rest of Asia Pacific
- Middle East & Africa
- Saudi Arabia
- UAE
- Israel
- South Africa
- Rest of Middle East & Africa
- Latin America
- Mexico
- Brazil
- Argentina
- Rest of Latin America
Single-Use Filtration Assemblies Market Report Scope
|
Report Attributes |
Details |
|
Market Size in 2025 |
USD 4.26 Billion |
|
Market Size in 2026 |
USD 5.05 Billion |
|
Revenue Forecast by 2034 |
USD 20.10 Billion |
|
CAGR |
18.9% from 2026 to 2034 |
|
Base Year |
2025 |
|
Historical Data |
2021–2024 |
|
Forecast Period |
2026–2034 |
|
Quantitative Units |
Revenue in USD Billion and CAGR from 2026 to 2034 |
|
Report Coverage |
Revenue Forecast, Competitive Landscape, Growth Factors, and Industry Trends |
|
Segments Covered |
|
|
Regional Scope |
|
|
Competitive Landscape |
|
|
Report Format |
|
|
Customization |
Report customization as per your requirements with respect to countries, regions, and segmentation. |
FAQ's
The global market size was valued at USD 4.26 Billion in 2025 and is projected to grow to USD 20.10 Billion by 2034.
North America dominated the market with 39.38% revenue share owing to robust manufacturing capabilities for biologics and high use of disposable bioprocess equipment.
Major applications include monoclonal antibody manufacturing, vaccine production, gene therapy, cell therapy, and buffer preparation.
A few of the key players in the market are Merck KGaA, Danaher Corporation, Sartorius AG, Thermo Fisher Scientific Inc., Parker-Hannifin Corporation, Repligen Corporation, Donaldson Company Inc., Meissner Filtration Products Inc., Asahi Kasei Corporation, 3M Company, Eaton Corporation plc, Avantor Inc., and others.
Key drivers include rising biologics demand, vaccine capacity expansion, and growing need for contamination control.
Key drivers include biopharmaceuticals, contract development and manufacturing organizations (CDMOs), vaccine producers, biotech companies, and research institutions.
Growth potential is favorable owing to increasing therapies and innovations in filtration technologies.
Download Sample Report of Single-Use Filtration Assemblies Market
Please fill out the form to request a customized copy of the research report.
