Peripheral Vascular Device Market Share, Size, Industry Analysis Report, 2026 - 2034
REPORT DETAILS
REPORT DETAILS
ABOUT THIS REPORT
Market Statistics
Peripheral Vascular Device Market Overview
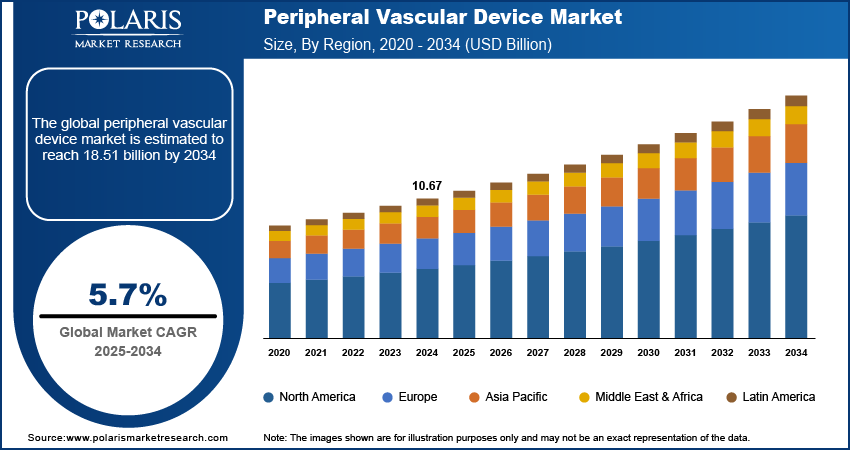
The global peripheral vascular device market size was valued at USD 11.26 billion in 2025. The market is projected to grow from USD 11.90 billion in 2026 to USD 18.56 billion by 2034, at a CAGR of 5.7% from 2026 to 2034. The growing existence of peripheral arterial disease (PAD) in entrenched and other vascular diseases are becoming more normal and geriatric population and other probabilities such as diabetes, obesity, and hypertension are causing the market to expand.
Key Takeaways
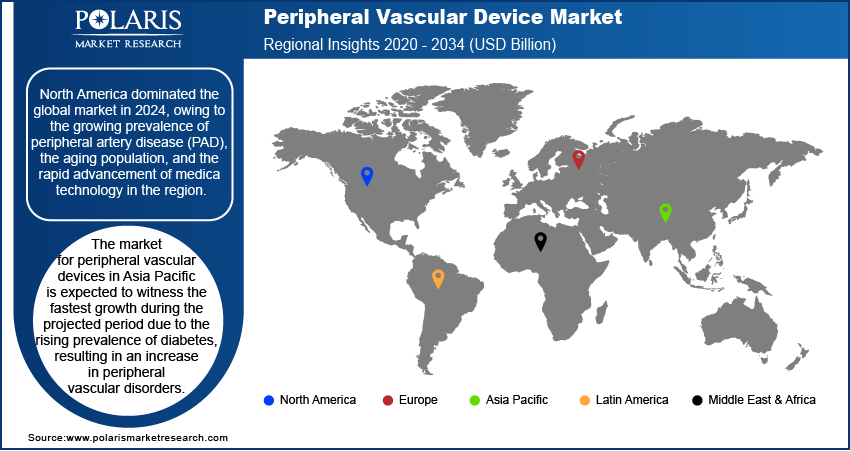
- North America peripheral vascular device market garnered the largest share valued at 38.54% in the region because of growing existence of PAD, the maturing population, and the speedy advancement and arrangement of progressive medical techniques.
- Asia Pacific is anticipated to grow at the fastest rate of 7.25%, due to the surging existence of diabetes, pushed by demographic moves and altering lifestyles, which has pushed the market forward.
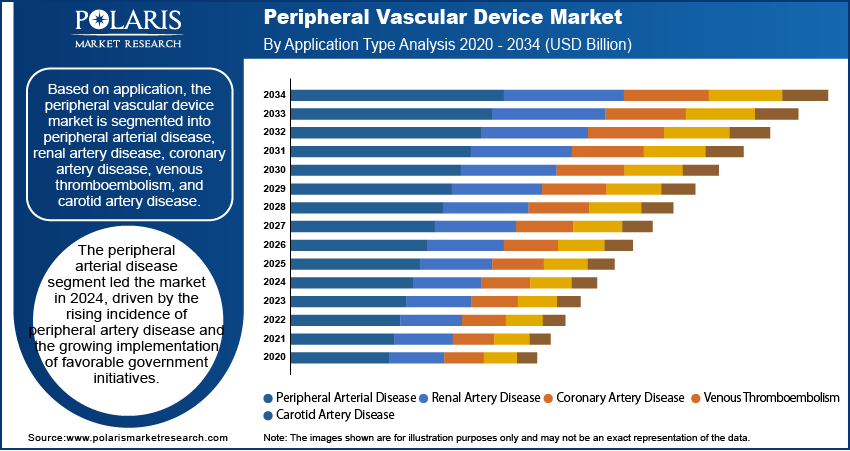
- The peripheral arterial disease segment witnessed the maximum revenue share of 36.48% in 2025.
- The stents segment accounted for 32.45% share of the market in 2025, due to its sizeable application in handling of peripheral artery disease.
- The catheters segment is the fastest growing segment and is expected to grow at a CAGR of 6.01% from 2026 to 2034. This is backed by rising use in minimally invasive processes and increasing adoption in diagnostic and therapeutic applications.
- The atherectomy devices segment held a revenue share of 13.28% in 2025. This share helped by its growing use in plaque removal procedures and increasing preference in complex peripheral artery disease cases.
*Note: Figures and projections outlined in this report are the result of Polaris Market Research’s proprietary analytical processes, grounded in the latest available datasets and market observations.
Industry Dynamics
- The growing existence of diabetes worldwide is propelling the market growth.
- Technological progressions have caused the advancement of convenient, less invasive and excessively productive peripheral vascular devices that boosts the market demand.
- The growing prevalence of emergency and crucial care acceptance for arterial blockage is anticipated to reinforce the market advancement in the near future.
- But recurring product fiasco and recalls and strict product approval procedures may impede the market growth.
Market Statistics
- 2025 Market Size: USD 11.26 billion
- 2034 Projected Market Size: USD 18.56 billion
- CAGR (2026-2034): 5.7%
- North America: Largest Market in 2025
To Understand More About this Research: Download Sample Report
What are Peripheral Vascular Devices?
Peripheral vascular devices are used to treat peripheral vascular illnesses, which are blood circulation issues caused by blockages, narrowing, or spasms in blood vessels due to the accumulation of fat and plaque. Vascular grafts, peripheral venous catheters, stents, and catheters are some commonly used peripheral vascular devices.
The rising prevalence of peripheral arterial disease (PAD) in established and emerging economies is driving the peripheral vascular device market growth. PAD and other vascular illnesses are becoming more common due to the aging population and other risk factors such as diabetes, obesity, and hypertension. The growing number of vascular health-focused clinical trials and research is also contributing to the market expansion. With clinical studies, researchers can assess the safety and effectiveness of novel tools and methods for treating PVD. These studies can also confirm current technologies and support the development of new treatments to cater to diverse medical needs.
Technological advancements in device design, including the development of minimally invasive treatments that use drug-eluting stents to reduce recovery time and complexities, are making peripheral vascular devices more appealing to patients and healthcare professionals. Additionally, the rising frequency of emergency and critical care admissions for arterial blockage treatment is expected to support the peripheral vascular device market development in the coming years.
Market Dynamics
Rising Incidence of Diabetes Globally
The growing prevalence of diabetes globally is propelling the peripheral vascular device market revenue. Diabetes damages blood vessels through a process called atherosclerosis, which involves the accumulation of plaque inside arteries; however, certain diabetes drugs, such as SGLT2 inhibitors and GLP-1 analogues, have been shown to help reduce cardiovascular risk and slow the progression of vascular damage. This process is significantly fueled by inflammation caused by high blood sugar levels associated with diabetes. Healthcare providers better address these complications with AI in diagnostics improving early detection and treatment planning. People with diabetes are at risk of developing PAD and other peripheral vascular disorders that mostly affect blood circulation in the lower limbs. With the growing diabetic population, PAD cases and severe outcomes such as severe limb ischemia and amputations are also rising. To address these issues, stents, angioplasty balloons, and atherectomy devices are essential for restoring blood flow.
Technological Advancements in Diagnostics
Technological advancements have led to the development of user-friendly, less intrusive, and highly effective peripheral vascular devices. PAD and other vascular illnesses can now be diagnosed and treated with greater accuracy due to advancements in modern imaging technology. Technological advancements in peripheral vascular devices include drug-coated balloons, stents, atherectomy devices, and wearable sensors. These devices are intended to improve treatment options and outcomes for peripheral artery disease (PAD). Thus, technological advancemnts are propelling the expansion of the peripheral vascular devices market globally.

Segment Insights
Evaluation by Application Insights
The global vascular device market, based on application, is segmented into peripheral arterial disease, renal artery disease, coronary artery disease, venous thromboembolism, and carotid artery disease. The peripheral arterial disease segment dominated the market with a 36.48% revenue share in 2025, driven by the rising incidence of peripheral artery disease and the growing implementation of favorable government initiatives. In addition, the increasing prevalence of cardiovascular disorders and improvements in diagnostic tools have led to a notable surge in PAD research in recent years, thereby contributing to the segment’s leading market position.
Assessment by Device Insights
Based on device, the market for peripheral vascular device market is segmented as catheters, atherectomy devices, stents, endograft, and stent-grafts. The stents segment of the peripheral device market dominated the market with 32.35%, in 2025 due to its extensive application in the management of peripheral artery disease. The rising incidence of cardiovascular illnesses is also driving the need for stents. In addition, technological developments in stents, including the development of drug-eluting stents, contribute to the segment’s leading position in the market.
The catheters segment is expected to grow at a CAGR of 6.01% during the forecast period. Catheters are widely used in therapeutic and diagnostic treatments, including atherectomy and angioplasty. Catheters allow access to peripheral arteries, enabling doctors to deliver treatment devices like stents, balloons, or embolization agents directly to the affected area. The growing prevalence of PAD, coupled with advancements in catheter technology like drug-eluting balloons and an increase in minimally invasive operations, is driving the market for peripheral vascular catheters.
Modality-Based Segmentation: Minimally Invasive vs Invasive Procedures
| Parameter | Minimally Invasive | Invasive |
| Procedure Type | Small incision or catheter-based procedures | Open surgical procedures |
| Recovery Time | Short recovery period | Longer recovery period |
| Risk Level | Lower risk of complications | Higher risk due to surgery |
| Hospital Stay | Short hospital stay or outpatient | Longer hospital stay |
| Patient Preference | High preference due to less pain and quick recovery | Lower preference except in severe cases |
| Cost Impact | Cost-effective in long term due to faster recovery | Higher overall cost due to surgery and hospitalization |
| Usage in Market | Increasing adoption with advanced technologies | Used in complex and critical conditions |
| Examples | Angioplasty, stenting, catheter-based treatments | Bypass surgery, open vascular repair |
Regional Analysis
By region, the report offers peripheral vascular device market insights into North America, Europe, Asia Pacific, Latin America, and the Middle East and Africa. North America led the global market with a 38.54% market share in 2025, driven by the increasing prevalence of PAD, the aging population, and the quick development and deployment of advanced medical technologies. The rising number of PAD patients highlights the growing need for advanced stenting methods to deal with the complexities of the disease. The introduction of new products, rising healthcare costs, and the presence of a robust healthcare infrastructure are other factors driving the regional market dominance.
The Asia Pacific peripheral vascular device market is anticipated to register the fastest growth at a CAGR of 7.25%, during the projected period. The rising prevalence of diabetes, driven by demographic shifts and changing lifestyles, has led to an increase in peripheral vascular illnesses across the Asia Pacific. Additionally, the development of new, minimally invasive vascular intervention devices due to continuous improvements in medical technology is having a favorable impact on the regional market growth. The improving healthcare infrastructure in emerging countries further contributes to the market expansion in the region.
Key Players and Competitive Insights
Leading market players are investing heavily in research and development to expand their offerings, which will help the market grow even more. Market participants are also undertaking a variety of strategic activities, such as innovative product launches, international collaborations, higher investments, and mergers and acquisitions to expand their global footprint. To expand and survive in a more competitive and rising market environment, market participants must offer cost-effective solutions.
The peripheral vascular device market faces competition from major global players. These players dominate the market with their extensive production capacities and advanced technologies. Competition is further heightened by fluctuating raw material prices, stringent environmental regulations, and the necessity for technological advancements to improve device efficiency. A few of the major key players involved in the market are Abbott Laboratories, Angioscore, Edward Lifesciences Corporation, Medtronic, Becton, Teleflex Medical, Volcano Corporation, BD, Teleflex Medical, Cook Group, Cordis Corporation, and St. Jude Medical.
List of Key Players
- Abbott Laboratories
- BD
- Becton
- C.R.Bard
- Cook Group
- Cordis Corporation
- Covidien
- Edward Lifesciences Corporation
- Medtronic
- St. Jude Medical
- Teleflex Medical
- Volcano Corporation
Future of Peripheral Vascular Device Market
The market is expected to grow due to the rising number of cardiovascular diseases and peripheral artery disease cases. Innovation in minimally invasive devices will continue to increase adoption across hospitals. Advancements in imaging and device technology will improve treatment outcomes and procedure accuracy. Improving healthcare infrastructure in developing regions will help market expansion. Demand will increase in emerging markets because of changing lifestyle and increasing patient number.
Peripheral Vascular Device Industry Developments
- March 2026: Abbott Laboratories introduced the XIENCE Skypoint stent in India. It is used for complex PCI procedures. It supports treatment of long lesions and large vessels. It can also be used in left main coronary artery cases. (Source: abbott.in)
- February 2026: Avantec Vascular introduced a 510(k)-cleared thrombectomy system for deep vein thrombosis (DVT). (Source: nipro-group.com)
- September 2025: Johnson & Johnson launched the Shockwave Javelin IVL catheter in Europe for heavily calcified lesions. (Source: jnj.com)
- July 2025: MicroPort Endovastec reported the first commercial use of its Talos Thoracic Stent Graft System in Argentina. The device was used to treat a Stanford type B aortic dissection with a single personalized stent graft, successfully restoring blood flow while reducing the risk of spinal cord injury. Source: microport.com)
- December 2024: Terumo Interventional Systems (TIS) launched its R2P NaviCross Peripheral Support Catheter. According to TIS, the new catheter comes with a double-braided construction to ensure enhanced trackability and torque control. (Source: terumo.com)
- October 2024: BD introduced its new BD Intraosseous Vascular Access System. The new system comes with an electric IO driver that allows for rapid access to the circulatory system in critical care situations. (Source: bd.com)
- June 2024: Philips introduced their Duo Venous Stent System in the US, targeting people with venous blockages. With premarket approval (PMA) from the US Food and Drug Association (FDA), this innovative device aims to address the root causes of chronic deep vein thrombosis. (Source: philips.com)
- December 2024: Abbott's launched Esprit BTK System, which makes use of an Enviroximes Eluting Resorbable Scaffold, was formally approved by the US FDA. According to Abbott, this innovative method is designed to treat chronic limb-threatening ischemia (CLTI) that affects the BTK. Source: abbott.com)
Peripheral Vascular Device Market Segmentation
By Device Outlook
- Catheters
- Atherectomy Devices
- Stents
- Endograft
- Stent-Grafts
By Material Outlook
- Biodegradable
- Nitinol-Based
- Metallic
- Polymer-Based
- Platinum-Based
By Application Outlook
- Peripheral Arterial Disease
- Renal Artery Disease
- Coronary Artery Disease
- Venous Thromboembolism
- Carotid Artery Disease
By Modality Outlook
- Minimally Invasive
- Invasive
- Non-Invasive
By Regional Outlook
- North America
- US
- Canada
- Europe
- Germany
- France
- UK
- Italy
- Spain
- Netherlands
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Malaysia
- South Korea
- Indonesia
- Australia
- Rest of Asia Pacific
- Middle East & Africa
- Saudi Arabia
- UAE
- Israel
- South Africa
- Rest of the Middle East & Africa
- Latin America
- Mexico
- Brazil
- Argentina
- Rest of Latin America
Report Scope
| Report Attributes | Details |
| Market Size Value in 2025 | USD 11.26 billion |
| Market Size Value in 2026 | USD 11.90 billion |
| Revenue Forecast by 2034 | USD 18.56 billion |
| CAGR | 5.7% from 2026 to 2034 |
| Base Year | 2025 |
| Historical Data | 2021–2024 |
| Forecast Period | 2026–2034 |
| Quantitative Units | Revenue in USD billion and CAGR from 2026 to 2034 |
| Report Coverage | Revenue Forecast, Market Competitive Landscape, Growth Factors, and Trends |
| Segments Covered |
|
| Regional Scope |
|
| Competitive Landscape |
|
| Report Format |
|
| Customization | Report customization as per your requirements with respect to countries, regions, and segmentation. |
FAQ's
The peripheral vascular device market was valued at USD 11.26 billion in 2025 and is projected to grow to USD 18.56 billion in 2034.
The market is projected to register a CAGR of 5.7% during the forecast period.
North America held the largest market share valued at 38.54% in 2025.
Becton, Teleflex Medical, Volcano Corporation, BD, Teleflex Medical, Cook Group, Cordis Corporation, Covidien, Edward Lifesciences, C.R.Bard, St. Jude Medical, Medtronic, and Abbott Laboratories are a few of the key players in the market.
The peripheral arterial disease segment led the market with 36.48% in 2025.
The stents segment held the largest market share accounting for 32.45% in 2025.
Download Sample Report of global peripheral vascular device market
Please fill out the form to request a customized copy of the research report.
