Viscosupplementation Market Size, Share, Analysis Report, 2025-2034
REPORT DETAILS
REPORT DETAILS
ABOUT THIS REPORT
Market Overview
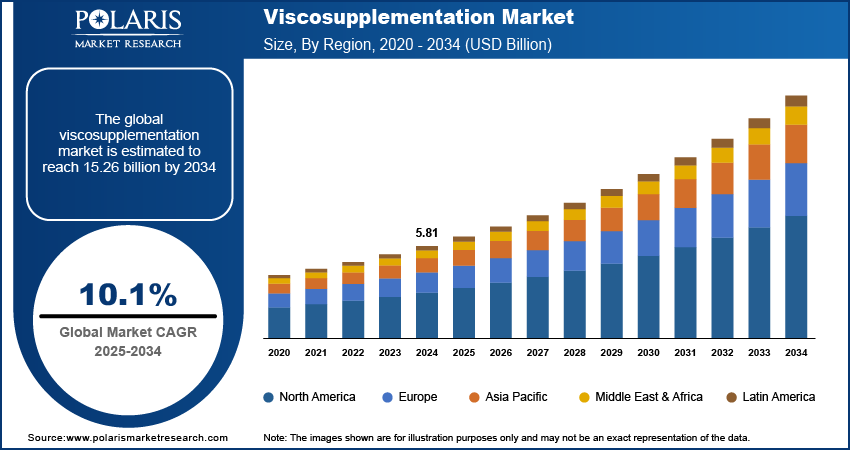
The global viscosupplementation market size was valued at USD 5.81 billion in 2024. The market is projected to grow from USD 6.39 billion in 2025 to USD 15.26 billion by 2034, exhibiting a CAGR of 10.1% from 2025 to 2034. Rising prevalence of osteoarthritis and joint disorders drives the market, as demand grows for minimally invasive treatments that improve joint lubrication, reduce pain, and enhance mobility, supporting better patient quality of life and reducing reliance on surgical interventions.
Key Insights
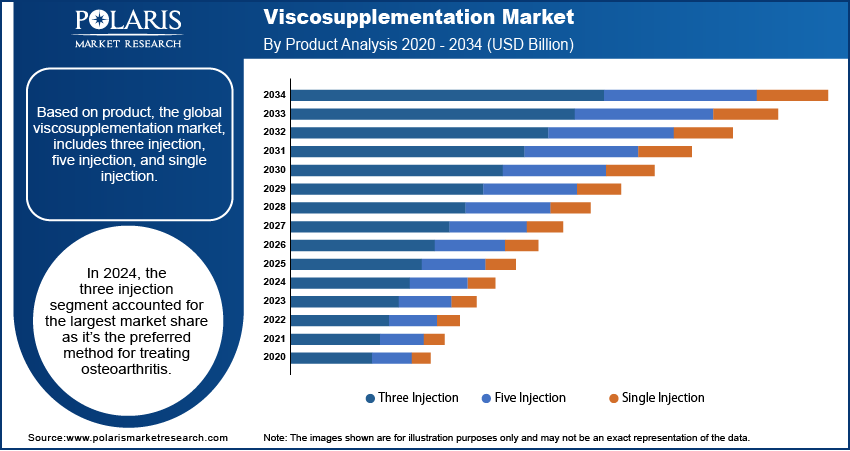
- In 2024, the three-injection segment held the largest market share, as it remains the favored approach for osteoarthritis treatment among healthcare professionals.
- From 2025 to 2034, the orthopedic clinics/ASCs segment is expected to grow the fastest, driven by rising demand for outpatient surgical procedures.
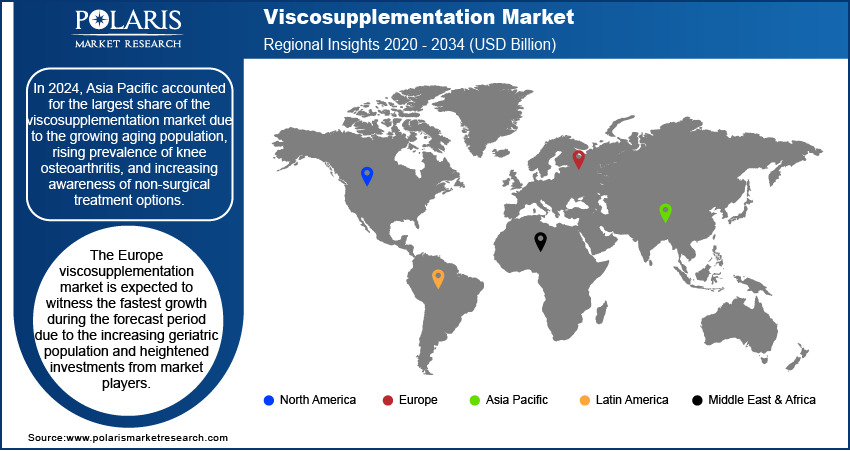
- Asia Pacific accounted for the largest share in 2024, supported by an expanding elderly population, higher knee osteoarthritis prevalence, and greater awareness of non-surgical therapies.
- The Europe viscosupplementation market is projected to experience the quickest growth during the forecast period, fueled by a growing geriatric population and increased investments from industry participants.
Industry Dynamics
- Increasing prevalence of osteoarthritis and joint-related disorders is driving strong demand for viscosupplementation treatments, offering pain relief and improved mobility across aging populations worldwide.
- Growing adoption of minimally invasive procedures and outpatient care is accelerating market growth by enhancing patient convenience and reducing healthcare costs.
- High treatment costs, limited insurance coverage, and variability in clinical outcomes continue to challenge widespread adoption and accessibility of viscosupplementation therapies globally.
- Advances in formulation technologies, including cross-linked hyaluronic acid and combination therapies, are improving treatment durability and patient satisfaction, driving innovation within the market.
Market Statistics
- 2024 Market Size: USD 5.81 billion
- 2034 Projected Market Size: USD 15.26 billion
- CAGR (2025-2034): 10.1%
- Asia Pacific: Largest market in 2024
To Understand More About this Research: Download Sample Report
Viscosupplementation is a minimally invasive medical procedure involving the direct injection of a gel-like fluid, composed of exogenous hyaluronic acid (HA) derivatives, into the knee joints. This fluid acts as a lubricant, facilitating smooth bone movement, reducing inflammation, and serving as a shock absorber for joint loads. Viscosupplementation offers comprehensive benefits by decreasing friction during movements, enhancing synovial fluid cushioning, alleviating severe pain, and improving joint range of motion. Additionally, it restores the rheological properties of synovial fluids, providing mechanical, analgesic, anti-inflammatory, and chondroprotective effects. As a result, viscosupplementation is extensively used in the treatment of osteoarthritis in the knee, hip, and hand, with formulations available in intermediate, low, and high molecular weights.
The growing preference for single-injection therapy for osteoarthritis is expected to drive market growth during the forecast period. Factors such as an aging population, smoking, a sedentary lifestyle, and other lifestyle changes are also contributing to the increased demand for osteoarthritis and osteoporosis treatments.
Market Dynamics
Rising Prevalence of Knee Osteoarthritis
Knee OA is one of the most common forms of arthritis, leading to significant pain, reduced mobility, and decreased quality of life for affected individuals. For instance, according to WHO, in 2023, approximately 73% of osteoarthritis patients are aged 55 and above, with females making up 60% of the affected population, highlighting age and gender as significant risk factors. The knee is the most affected joint, with 365 million cases, followed by the hip and hand. The condition’s prevalence is increasing due to the aging global population, along with lifestyle factors such as obesity, physical inactivity, and the growing incidence of chronic diseases like hypertension and diabetes, all of which contribute to the deterioration of joint health. As a result, there is a growing demand for effective treatments that can alleviate pain and improve joint function. Viscosupplementation, which involves injecting hyaluronic acid into the knee joint to restore lubrication and reduce inflammation, has emerged as a popular therapeutic option. This treatment provides a non-surgical alternative for patients with mild to moderate knee OA who seek pain relief and improved mobility. The growing awareness of viscosupplementation’s effectiveness, coupled with the increasing number of patients seeking relief from knee OA, positions the viscosupplementation market for continued growth.
Growing Aging Population Worldwide
The prevalence of age-related conditions, such as knee osteoarthritis (OA), is rising as the global population ages. Older adults are more susceptible to joint degeneration, contributing to a higher incidence of knee OA, which is a leading cause of chronic pain and disability in this demographic such patients require prothetics and orthotics. As the aging population increasingly seeks non-invasive solutions to manage their symptoms, viscosupplementation has become a preferred treatment option. Advancements in the formulation of hyaluronic acid, along with its proven efficacy in improving joint function, have made viscosupplementation an attractive alternative to surgery, especially for elderly patients. Thus, the growing aging population is a key factor driving the market expansion.

Segment Insights
By Product Outlook
The global viscosupplementation market, based on product, is bifurcated into three injection, five injection, and single injection. In 2024, the three injection segment accounted for the largest market share, as it is the preferred method for treating osteoarthritis among medical practitioners. This approach is often favored as it reduces the need for total knee replacement surgery in osteoarthritis cases. The segment’s dominance is also influenced by the availability of various three-injection options. Additionally, numerous clinical trials evaluating the efficacy of three-injection viscosupplementation have been approved, showing positive results for its use in the treatment of osteoarthritis
By End Use Outlook
The global viscosupplementation market, based on end use, is segmented into orthopedic clinics/ASCs, and hospitals. The orthopedic clinics/ASCs segment is projected to experience the fastest growth from 2025 to 2034 due to the increased demand for outpatient surgeries. Orthopedic clinics specialize in the care of the musculoskeletal system, addressing both surgical and nonsurgical issues related to the spine, joints, ligaments, bones, muscles, and nerves enhancing further orthopedic devices. The growing prevalence of osteoarthritis among older people is a key factor expected to drive growth of this segment. Additionally, ambulatory surgical centers (ASCs), which efficiently conduct same-day surgeries for procedures like ACL surgery, hip, knee, and others, are contributing to this trend. These surgeries are relatively uncomplicated and often require minimal hospitalization. ASCs enable orthopedic clinics to accommodate a higher volume of patients daily. According to the Global RA Network, the global prevalence of arthritis surpassed 350 million people in 2021 and continues to rise, primarily due to the aging population. Additionally, hospitals are expected to experience sustained growth in the coming years, fueled by advancements in medical technology and the development of multispecialty hospitals.
Regional Analysis
By region, the study coversNorth America, Europe, Asia Pacific, Latin America, and the Middle East & Africa. In 2024, Asia Pacific accounted for the largest share of the global population due to the growing aging population, rising prevalence of knee osteoarthritis, and increasing awareness of non-surgical treatment options. Countries such as China, Japan, and India have witnessed significant demand for viscosupplementation due to their large elderly populations and higher incidences of osteoarthritis. Additionally, improvements in healthcare infrastructure, government support for affordable treatments, and the availability of advanced hyaluronic acid-based products have contributed to market growth. The expanding middle class and increasing disposable income across the Asia Pacific further enhance access to these treatments, making it the dominant region in the viscosupplementation market.
The Europe viscosupplementation market is expected to witness the fastest growth during the forecast period due to the increasing geriatric population and heightened investments from market players. For instance, according to the International Osteoporosis Foundation, in October 2023, 60.9% of women over 50 in the UK reported fractures from minor falls, while 51.1% of non-fracture cases had never discussed bone health with their doctors. About 43.7% underwent diagnostic scans, highlighting gaps in osteoporosis awareness and treatment. These factors emphasize the rising demand for effective joint care solutions, driving the adoption of viscosupplementation procedures to improve mobility and reduce pain in aging populations.
Key Players and Competitive Insights
The market is highly competitive, with both global and regional players striving to strengthen their positions through innovation, partnerships, and strategic expansions. Leading companies are focusing on developing advanced hyaluronic acid formulations, including single-injection and cross-linked variants, to enhance efficacy and patient compliance. Key players, such as Sanofi, Fidia Farmaceutici, Seikagaku Corporation, and Anika Therapeutics, are leveraging research and development to maintain a competitive edge. Collaborations with healthcare providers and distributors, along with robust marketing strategies, are common tactics to expand market reach. Favorable reimbursement policies in regions like Europe and increasing regulatory approvals in emerging markets further intensify the competition. Additionally, smaller players are gaining traction by offering cost-effective solutions, especially in price-sensitive markets like Asia Pacific and Latin America.
The rising adoption of minimally invasive surgeries and the growing prevalence of knee osteoarthritis provide opportunities for market expansion with penetration of orthopedic surgical robots as well. However, challenges such as pricing pressure, stringent regulations, and competition from alternative treatments like corticosteroid injections and platelet-rich plasma therapy compel companies to continually innovate to maintain market relevance. The competitive landscape of the market remains dynamic, driven by technological advancements and an increasing focus on patient-centric care. A few key market players are Anika Therapeutics, Inc.; DePuy Synthes; F. Hoffmann-La Roche Ltd.; Ferring Pharmaceuticals B.V.; Fidia Farmaceutici S.p.A, LG Life Sciences Ltd.; Lifecore Biomedical; Sanofi; Seikagaku Corp.; Smith & Nephew PLC; and Zimmer Biomet.
Johnson & Johnson Services, Inc. (DePuy Synthes) is a multinational corporation based in New Brunswick, New Jersey, USA. The company operates through pharmaceuticals, medical devices, and consumer health products segments. Its subsidiary, Janssen Pharmaceuticals, Inc., develops and markets drugs in various therapeutic areas, including neuroscience, oncology, infectious diseases, and immunology. Janssen Pharmaceuticals, Inc. is known for creating many well-established drugs such as Risperdal (risperidone), Remicade (infliximab), and Zytiga (abiraterone).
Sanofi is a healthcare company that researches, develops, manufactures, and markets therapeutic solutions worldwide. It operates through three segments: pharmaceuticals, vaccines, and consumer healthcare. In the pharmaceuticals segment, Sanofi provides specialty care treatments for various conditions, including dupixent, rare diseases, oncology, neurology and immunology, rare blood disorders, and medicines for diabetes and cardiovascular diseases. The vaccines segment offers pediatric and travel vaccines, such as influenza, meningitis, and hepatitis A. The consumer healthcare segment provides over-the-counter products for cough, cold, flu, allergies, pain, and various wellness needs. Sanofi has collaborations and license agreements with several biopharmaceutical companies to develop novel therapies, including small-molecule drugs for oncology and immunology, treatments for Parkinson's disease, antibody-drug conjugates for cancer, and genome editing technologies. The company also collaborates to discover and develop small molecules, IgM antibodies, T-cell engagers, and cytokine therapies
List of Key Companies
- Anika Therapeutics, Inc.
- DePuy Synthes
- F.Hoffmann-La Roche Ltd.
- Ferring Pharmaceuticals B.V.
- Fidia Farmaceutici S.p.A
- LG Life Sciences Ltd.
- Lifecore Biomedical
- Sanofi
- Seikagaku Corp.
- Smith & Nephew PLC
- Zimmer Biomet
Industry Developments
In July 2024, LG Chem announced that its partner, Yifan Pharmaceutical, has launched Synovian in China. Synovian is a single-dose intra-articular treatment for osteoarthritis, marketed under the name Hyruan One.
In November 2022, Anika Therapeutics, Inc., a prominent joint preservation company, revealed it effectively met the primary endpoint in the Phase III Study for Cingal. This medication is formulated to offer prolonged pain relief for individuals dealing with knee osteoarthritis (OA). The company is focused on securing FDA clearance for Cingal and its introduction to the United States.
In April 2022, Juniper Biologics and Kolon Life Sciences entered into a licensing arrangement to develop and commercialize TissueGene-C low dose (TG-C LD), an investigational therapy tailored for non-surgical treatment through a single intra-articular injection, specifically addressing knee osteoarthritis. As per the terms of the licensing agreement, Juniper will lead the efforts in developing and promoting this gene therapy, with a focus on medical professionals and hospitals in the Asia Pacific, Middle East, and Africa.
Market Segmentation
By Product Outlook (Revenue – USD Billion, 2020–2034)
- Three Injection
- Five Injection
- Single Injection
By End Use Outlook (Revenue – USD Billion, 2020–2034)
- Orthopedic Clinics/ASCs
- Hospitals
By Regional Outlook (Revenue – USD Billion, 2020–2034)
- North America
- US
- Canada
- Europe
- Germany
- France
- UK
- Italy
- Spain
- Netherlands
- Russia
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Malaysia
- South Korea
- Indonesia
- Australia
- Vietnam
- Rest of Asia Pacific
- Middle East & Africa
- Saudi Arabia
- UAE
- Israel
- South Africa
- Rest of Middle East & Africa
- Latin America
- Mexico
- Brazil
- Argentina
- Rest of Latin America
Report Scope
|
Report Attributes |
Details |
|
Market Size Value in 2024 |
USD 5.81 billion |
|
Market Size Value in 2025 |
USD 6.39 billion |
|
Revenue Forecast by 2034 |
USD 15.26 billion |
|
CAGR |
10.1% from 2025 to 2034 |
|
Base Year |
2024 |
|
Historical Data |
2020–2023 |
|
Forecast Period |
2025–2034 |
|
Quantitative Units |
Revenue in USD billion and CAGR from 2025 to 2034 |
|
Report Coverage |
Revenue Forecast, Market Competitive Landscape, Growth Factors, and Trends |
|
Segments Covered |
|
|
Regional Scope |
|
|
Competitive Landscape |
|
|
Report Format |
|
|
Customization |
Report customization as per your requirements with respect to countries, regions, and segmentation. |
FAQ's
The global market was valued at USD 5.81 billion in 2024 and is projected to grow to USD 15.26 billion by 2034.
The global market is projected to register a CAGR of 10.1% from 2025 to 2034.
In 2024, Asia Pacific accounted for the largest share of the viscosupplementation market due to the growing aging population, rising prevalence of knee osteoarthritis, and increasing awareness of non-surgical treatment options.
A few key players in the market are Anika Therapeutics, Inc.; DePuy Synthes; F. Hoffmann-La Roche Ltd.; Ferring Pharmaceuticals B.V.; Fidia Farmaceutici S.p.A, LG Life Sciences Ltd.; Lifecore Biomedical; Sanofi; Seikagaku Corp.; Smith & Nephew PLC; and Zimmer Biomet.
In 2024, the three injection segment accounted for the largest market share as it’s the preferred method for treating osteoarthritis.
The orthopedic clinics/ASCs segment is projected to experience the fastest growth during the forecast period due to the increased demand for outpatient surgeries.
Download Sample Report of Viscosupplementation Market
Please fill out the form to request a customized copy of the research report.
